|
|
THE PEST CONTROL PRODUCTS (LICENSING OF PREMISES) REGULATIONS
ARRANGEMENT OF REGULATIONS
| 2. |
Premises not to be used unless licensed
|
| 3. |
Application for licensing of premises
|
| 4. |
Issue and expiry of licence
|
| 5. |
Layout and construction of manufacturing, formulating, packaging and storing premises
|
| 6. |
Protection of workers and operators and quality of products
|
| 7. |
Essential requirements for operations, etc.
|
SCHEDULES
THE PEST CONTROL PRODUCTS (DISPOSAL) REGULATIONS, 2006
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Disposal) Regulations, 2006.
|
| 2. |
Disposal of pest control products for commercial purpose
| (1) |
No person shall dispose of any pest control product or their containers in a manner that shall be detrimental to man, animal and the environment.
|
| (2) |
No person shall carry out any disposal of pest control product or containers for commercial purposes unless that person or institution is in possession of a licence under these Regulations and all provisions of the Act have been complied with.
|
| (3) |
The use of any pest control product disposal method shall be approved by the Board subject to specific requirements laid down by the Board.
|
| (4) |
The transboundary movements of pesticides for disposal shall be subject to ratified conventions, protocols, treaties or agreements to which Kenya is a party.
|
| (5) |
Without prejudice to the provisions of the Environmental Management and Co-ordination Act, any person or institution desiring a licence for disposal of pest control products for commercial purposes shall apply to the Board for a licence in the prescribed Form A set out in the Schedule and shall be accompanied by the prescribed fees.
|
|
| 3. |
Application for disposal licence
| (1) |
The Board may approve an application made under Regulation 2 and shall issue a licence as set out in Form B in the Schedule if it is satisfied that:
| (a) |
the application contains the information required under these regulations;
|
| (b) |
approved methods of disposal would be used or applied;
|
| (c) |
the applicant has sufficient expertise to dispose of the pest control products in question.
|
|
| (2) |
Disposal for commercial purposes shall be supervised by the Board which shall issue a certificate of disposal in the prescribed Form C in the Schedule after successful disposal.
|
| (3) |
Where the Board does not approve an application it shall not be obliged to give any reason thereof.
|
| (4) |
The Board may attach to a licence such conditions as it deems fit.
|
| (5) |
No person or institution to which a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence to any person or institution without the approval of the Board, which approval shall be endorsed on the licence.
|
|
| 4. |
Cancellation or suspension of licence
| (1) |
The Board may cancel or suspend for a period it deems fit the licence issued under these Regulations if satisfied that-
| (a) |
the licensee, his servant or agent has been convicted of an offence under the Act; or
|
| (b) |
the licensee, his servant or agent has breached any of the terms or conditions of the licence; or
|
| (c) |
for any other reason, that it is in public interest to do so.
|
|
| (2) |
Before cancelling or suspending a licence under this regulation, the Board shall give the licensee twenty-one days' notice to show cause why the licence should not be cancelled or suspended and the Board's decision in the matter shall be final.
|
|
THE PEST CONTROL PRODUCTS (LICENSING OF PREMISES) REGULATIONS, 1984
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Licensing of Premises) Regulations, 1984.
|
| 2. |
Premises not to be used unless licensed
| (1) |
No person shall use any premises, or being the owner or occupier thereof permit or allow the premises to be used, for the purposes of manufacturing, formulating, packaging, selling or storing pest control products unless that person is in possession of a licence issued under these Regulations in respect of those premises.
|
| (2) |
No person shall operate any business in pest control unless that person is in possession of a license issued under these regulations in respect of that business.
|
| (3) |
No licence shall be issued under these Regulations as set out in Form C in the Schedule unless the Board is satisfied that the provisions of these Regulations have been complied with.
[L.N. 124/2006, r. 2.]
|
|
| 3. |
Application for licensing of premises
| (1) |
An application for a licence under these Regulations shall be in Form A in the Schedule.
|
| (2) |
An applicant for a licence shall, on request by the Board, supply any information that may be required for the purposes of these Regulations.
|
|
| 4. |
Issue and expiry of licence
| (1) |
Every licence issued under these Regulations shall be in Form B in the Schedule, and shall expire on the 31st December next following the date of issue.
|
| (2) |
No person to whom a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence without the approval of the Board, which approval shall be endorsed on the licence.
|
| (3) |
No licence shall be transferred from the premises in respect of which it was issued.
|
| (4) |
A licence issued by the Board under regulation 4(1) may be cancelled, suspended or revoked by the Board if the holder of such a licence contravenes the provisions of the Act or the Regulations made under it.
|
| (5) |
The information on licence cancellation, suspension or revocation under 4(4) shall be made known to the holder in writing and the general public by Gazette notice.
[L.N. 124/2006, r. 3.]
|
|
| 5. |
Layout and construction of manufacturing, formulating, packaging and storing premises
All premises used for the manufacturing, formulating, packaging and storing of pest control products shall—
| (a) |
be of a suitable design, layout and construction to ensure the health of workers and to avoid contamination of the environment;
|
| (b) |
have sufficient space for the placement of equipment and storage of materials which is necessary for the health of workers and operators;
|
| (c) |
have separate areas, either by partition, location or other effective means, for those operations which do not require workers to be exposed to pest control products.
|
|
| 6. |
Protection of workers and operators and quality of products
Every person who owns, operates or is in charge of premises used for the manufacturing, formulating, packaging and storing of pest control products shall ensure that—
| (a) |
| (i) |
the persons working in the premises wear adequate protective clothing; |
| (ii) |
the premises are well supplied with first-aid facilities to the satisfaction of the Board to cater for accidental poisoning; |
| (iii) |
the general health of the persons working on the premises is adequately catered for; and |
|
| (b) |
the quality of the products are within the prescribed limits.
|
|
| 7. |
Essential requirements for operations, etc.
Every person who owns, operates or is in charge of—
| (a) |
premises in which the manufacturing, formulating, packaging and storing of a pest control product is undertaken shall have adequate knowledge of the chemistry, toxicology, efficacy and general use of the product being dealt with to the satisfaction of the Board;
|
| (b) |
premises licensed under these Regulations shall ensure that the products are packaged and labeled as required by the provisions of this Act and the Regulations and shall keep stock records for a minimum of five years;
|
| (c) |
pest control product stores and other dispensing premises shall have adequate knowledge of efficacy, uses and handling precautions of all pest control products within the premises to the satisfaction of the Board;
|
| (d) |
premises licensed under these Regulations shall provide adequate water and general cleanliness to facilitate safe dispensing of the products; ensure physical separation of pest control products stores from food stores and keep stock records for all pest control products in the premises.
|
|
| 8. |
Permits
| (1) |
Any person who intends to handle, use, distribute, transport or deal in a Pest Control Product under restricted class shall apply to the Board for a permit in the prescribed Form D in the Schedule.
|
| (2) |
No person shall handle, use, distribute, transport or deal in a Pest Control Product under restricted class without a permit from the Board in the prescribed Form E in the Schedule.
[L.N. 124/2006, r. 7.]
|
|
THE PEST CONTROL PRODUCTS (LICENSING OF PREMISES) REGULATIONS
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Licensing of Premises) Regulations.
|
| 2. |
Premises not to be used unless licensed
| (1) |
No person shall use any premises, or being the owner or occupier thereof permit or allow the premises to be used, for the purposes of manufacturing, formulating, packaging, selling or storing pest control products unless that person is in possession of a licence issued under these Regulations in respect of those premises.
|
| (2) |
No person shall operate any business in pest control unless that person is in possession of a license issued under these regulations in respect of that business.
|
| (3) |
No licence shall be issued under these Regulations as set out in Form C in the Schedule unless the Board is satisfied that the provisions of these Regulations have been complied with.
[L.N. 124/2006, r. 2.]
|
|
| 3. |
Application for licensing of premises
| (1) |
An application for a licence under these Regulations shall be in Form A in the Schedule.
|
| (2) |
An applicant for a licence shall, on request by the Board, supply any information that may be required for the purposes of these Regulations.
|
|
| 4. |
Issue and expiry of licence
| (1) |
Every licence issued under these Regulations shall be in Form B in the Schedule, and shall expire on the 31st December next following the date of issue.
|
| (2) |
No person to whom a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence without the approval of the Board, which approval shall be endorsed on the licence.
|
| (3) |
No licence shall be transferred from the premises in respect of which it was issued.
|
| (4) |
A licence issued by the Board under regulation 4(1) may be cancelled, suspended or revoked by the Board if the holder of such a licence contravenes the provisions of the Act or the Regulations made under it.
|
| (5) |
The information on licence cancellation, suspension or revocation under 4(4) shall be made known to the holder in writing and the general public by Gazette notice.
[L.N. 124/2006, r. 3.]
|
|
| 5. |
Layout and construction of manufacturing, formulating, packaging and storing premises
All premises used for the manufacturing, formulating, packaging and storing of pest control products shall—
| (a) |
be of a suitable design, layout and construction to ensure the health of workers and to avoid contamination of the environment;
|
| (b) |
have sufficient space for the placement of equipment and storage of materials which is necessary for the health of workers and operators;
|
| (c) |
have separate areas, either by partition, location or other effective means, for those operations which do not require workers to be exposed to pest control products.
|
|
| 6. |
Protection of workers and operators and quality of products
Every person who owns, operates or is in charge of premises used for the manufacturing, formulating, packaging and storing of pest control products shall ensure that—
| (a) |
| (i) |
the persons working in the premises wear adequate protective clothing; |
| (ii) |
the premises are well supplied with first-aid facilities to the satisfaction of the Board to cater for accidental poisoning; |
| (iii) |
the general health of the persons working on the premises is adequately catered for; and |
|
| (b) |
the quality of the products are within the prescribed limits.
|
|
| 7. |
Essential requirements for operations, etc.
Every person who owns, operates or is in charge of—
| (a) |
premises in which the manufacturing, formulating, packaging and storing of a pest control product is undertaken shall have adequate knowledge of the chemistry, toxicology, efficacy and general use of the product being dealt with to the satisfaction of the Board;
|
| (b) |
premises licensed under these Regulations shall ensure that the products are packaged and labeled as required by the provisions of this Act and the Regulations and shall keep stock records for a minimum of five years;
|
| (c) |
pest control product stores and other dispensing premises shall have adequate knowledge of efficacy, uses and handling precautions of all pest control products within the premises to the satisfaction of the Board;
|
| (d) |
premises licensed under these Regulations shall provide adequate water and general cleanliness to facilitate safe dispensing of the products; ensure physical separation of pest control products stores from food stores and keep stock records for all pest control products in the premises.
|
|
| 8. |
Permits
| (1) |
Any person who intends to handle, use, distribute, transport or deal in a Pest Control Product under restricted class shall apply to the Board for a permit in the prescribed Form D in the Schedule.
|
| (2) |
No person shall handle, use, distribute, transport or deal in a Pest Control Product under restricted class without a permit from the Board in the prescribed Form E in the Schedule.
[L.N. 124/2006, r. 7.]
|
|
SCHEDULE
FORMS
|
FORM A
|
(r. 3)
|
|
THE PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
PEST CONTROL PRODUCTS (LICENSING OF PREMISES AND BUSINESS) (AMENDMENT) REGULATIONS
|
|
APPLICATION FOR A LICENCE
|
The Managing Director,
Pest Control Products Board,
Waiyaki Way,
P.O. Box 13794-00800,
Westlands,
NAIROBI.
| 1 |
Name and address of applicant/s ...................................................................
|
| 2 |
Name and address of persons, firm or company to be issued with licence
.................................................................................................................
|
| 3. |
Full names of partners and/or directors (Where applicable)
.................................................................................................................
|
| 4. |
Nature of occupation for which a licence is required
.................................................................................................................
|
| 5. |
Owner of premises ........................................................................................
|
| 6. |
Plot No. ....................................... L.R. No. .........................................
|
| 7. |
Location ................................... Road/Street .........................................
|
| 8. |
Town ......................................... District ...........................................
|
| 9. |
General business postal address .......................................................................
|
| 10. |
Name and qualifications of the person under whom processing/trading will be undertaken ................................................................................................................
|
|
|
|
Fee paid ......................
|
Date ...............................................
|
|
Date ..........................................
|
..................................................Signature of applicant
|
|
|
|
|
FORM B
|
(r. 4(1))
|
|
THE PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
PEST CONTROL PRODUCTS (LICENSING OF PREMISES AND BUSINESS) (AMENDMENT) REGULATIONS
|
|
Receipt No. ................................................
|
Licence No. ................................................
|
|
This licence is granted to .................................................................... to display /retail/wholesale/store/package manufacture/formulate/ pest control products at Plot L.R. No. ..................................................
|
|
Situated at ...............................................................................................................
|
|
Situated at ...............................................................................................................
|
|
............................................................Managing Director,
Pest Control Products Board
|
...............................................................Inspector,
Pest Control products Board
|
|
Date .................................................
|
Date .....................................................
|
|
This licence is not transferable to any other person without the approval of the Board.
|
|
THIS LICENCE IS NOT TRANSFERABLE TO ANY OTHER PREMISES
|
|
|
|
_______________________
|
|
FORM C
|
(r. 2(3))
|
|
THE PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
PEST CONTROL PRODUCTS (LICENSING OF PREMISES AND BUSINESS) (AMENDMENT) REGULATIONS
|
A. Inspection of Premises
During inspection of pest control products premises, the following items shall be examined and comments made in order that the correct action can be taken with regard to licensing. A copy of the inspection report shall be left with the owner or the person incharge of the premises. The original shall be returned to the Pest Control Products Board.
| 1. |
Name and address of Applicant/Business .......................................................................
|
| 2. |
Nature of Business ............................................................................................
|
| 3. |
Location of Premises ...........................................................................................
|
| 4. |
L.R. No./Plot No. ..............................................................................
B. Specific Requirements
|
ITEM
|
COMPLIANCE WITH REQUIREMENTS
|
INSPECTOR'S RECOMMENDATION TO OWNER
|
|
|
PHYSICAL FACILITIES
|
YES
|
NO
|
|
|
1.
|
Suitable design, layout and construction
|
|
|
|
|
2.
|
Sufficient space
|
|
|
|
|
|
PROTECTION FOR WORKERS
|
|
3.
|
Separate areas for workers who are not handling pesticides
|
|
|
|
|
4.
|
Protective clothing
|
|
|
|
|
5.
|
First-Aid Box/Antidotes
|
|
|
|
|
6.
|
Fire Extinguisher
|
|
|
|
|
7.
|
Adequate water within premises
|
|
|
|
|
8.
|
Floor - smooth concrete
|
|
|
|
|
9.
|
Storage and stacking of containers
|
|
|
|
|
10.
|
Aeration
|
|
|
|
|
11.
|
Sawdust and sumps for disposal of spills
|
|
|
|
|
12.
|
General Cleanliness
|
|
|
|
|
|
PROTECTION OF THE PUBLIC AND MAINTENANCE OF PRODUCT EFFICACY
|
|
13.
|
Physical separation of food and feeds from pesticides
|
|
|
|
|
14.
|
All products must be labelled as required by law
|
|
|
|
|
15.
|
All labels should show date of manufacture and shelve life
|
|
|
|
|
16.
|
Stock records kept
|
|
|
|
|
17.
|
Dustbin for disposal
|
|
|
|
|
18.
|
Qualified operators for manufacturing and formulating premises
|
|
|
|
|
19.
|
Qualified operators for store and dispensing premises.
|
|
|
|
C. General Recommendations
I certify that I have inspected the premises described above and it is my considered opinion that a licence should/should not be issued/withdrawn for the premises.
The following defects should be rectified (specify items by numbers)..........................
|
1. ...........................................................Signature of inspector
|
1. .....................................................Signature of Inspector
|
|
2. .........................................................Name of Inspector
|
2. .....................................................Name of inspector
|
|
|
FORM D
|
(r. 8(1))
|
|
PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
PEST CONTROL PRODUCTS (LICENSING OF PREMISES AND BUSINESS) (AMENDMENT) REGULATIONS
|
|
APPLICATION FOR PERMIT TO DEAL IN PEST CONTROL PRODUCTS UNDER RESTRICTED CLASS
|
Name and address of applicant ............................................................................
Status and address of applicant (Manufacture, Agent, Registrant, Distributor, User)
.........................................................................................
Name and address of registrant of pest control product
.................................................................................................................
Trade name, Formulation, Concentration ................................................................
Registration No. .......................................................................................
Target pest/diseases ....................................................................................
Package type ...........................................................................................
Quality .................................................................................................
Signature of applicant ..................................................................................
Date ...............................................
|
FORM E
|
|
(r. 8 (2)
|
|
PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
PERMIT No.
|
|
THE PEST CONTROL PRODUCTS (LICENSING OF PREMISES AND BUSINESS) (AMENDMENT) REGULATIONS
|
|
PERMIT TO DEAL IN PEST CONTROL PRODUCTS UNDER RESTRICTED CLASS
|
|
|
|
PERMIT No. .............................................
|
This permit is granted to ..............................................................................
To sell/store/transport/use product ....................................................................
Registration Number ........................ At plot number ..................................
L.R. No. ....................... Situated .........................................
|
..........................................................Managing Director
Pest Control Products Board
|
Date ......................................
|
THE PEST CONTROL PRODUCTS (REGISTRATION) REGULATIONS
ARRANGEMENT OF REGULATIONS
| 3. |
Exemption from registration
|
| 4. |
Application for registration of pest control products
|
| 4A. |
Application for registration of a synthetic or conventional pest control product
|
| 4B. |
Application for the registration of microbial biopesticide
|
| 4C. |
Application for the registration of macrobial biopesticide
|
| 4D. |
Application form for the registration of a biochemical pesticide other than semiochemicals
|
| 4E. |
Application form for registration of spray adjuvants
|
| 4F. |
Registration of a parallel/daughter pest control product
|
| 4G. |
Extension of the use of a registered pest control product
|
| 5. |
Applicant to provide samples
|
| 7. |
Issue of certificate of registration
|
| 8. |
Duration and renewal of certificate of registration
|
| 9. |
Temporary registration
|
| 10. |
Refusal to register pest control
|
| 11. |
Suspension and revocation of certificates of registration
|
| 12. |
Notice to holder of certificate of registration, etc.
|
| 15. |
Declared source of product
|
SCHEDULES
| FIRST SCHEDULE [r. 3] — |
ITEMS EXEMPTED FROM REGISTRATION
|
| SECOND SCHEDULE [r. 4] — |
FORMS
|
THE PEST CONTROL PRODUCTS (LICENCE FEES AND OTHER CHARGES) REGULATIONS, 2006
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Licence Fees and Other Charges) Regulations, 2006.
|
| 2. |
Fees payable
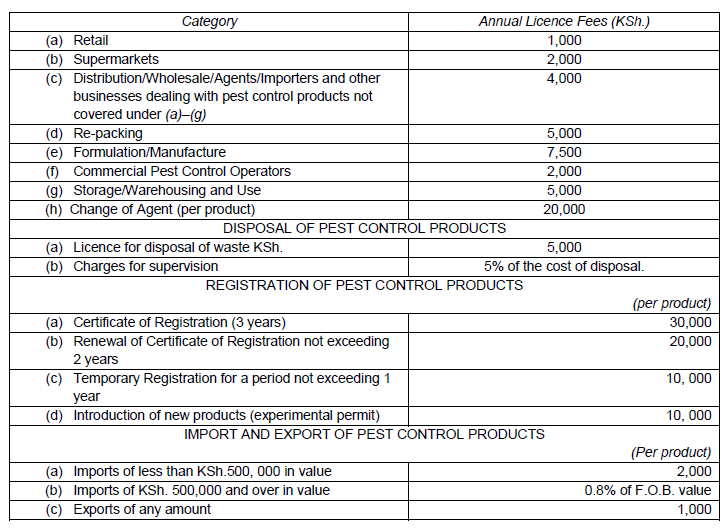
The fees payable to the Board by any person for services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule and the fees chargeable shall be added in case of combination of businesses in category I in the Schedule.
|
| 3. |
Charges for supervision services
The charges for supervision services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule.
|
| 4. |
Import fee
The Board may charge an import fee at the rate of 0.8% free on board (F.O.B.) value of the product and shall be paid to the Board as agreed with the Agrochemicals Association of Kenya of which 0.4% shall be used by the Board to train end-users and other stake holders on safe use of products and the remaining 0.4% shall be remitted by the Board to the Association for the same purpose of training.
|
SCHEDULE
LICENSING OF PREMISES
THE PEST CONTROL PRODUCTS (LABELLING, ADVERTISING AND PACKAGING) REGULATIONS, 1984
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Labelling, Advertising and Packaging) Regulations, 1984.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"display panel" means part of a label applied on or affixed to the package of a pest control product but does not include a leaflet or brochure unless it is part of the label;
"Formulator" means a person who prepares the active ingredient of a pest control product into a form which is convenient for its intended use;
"Re-packer" means a person who puts a formulated product into appropriate unit sizes.
[L.N. 127/2006, r. 2.]
|
| 3. |
Labelling requirements
| (1) |
No pest control product shall be distributed or sold without a label.
|
| (2) |
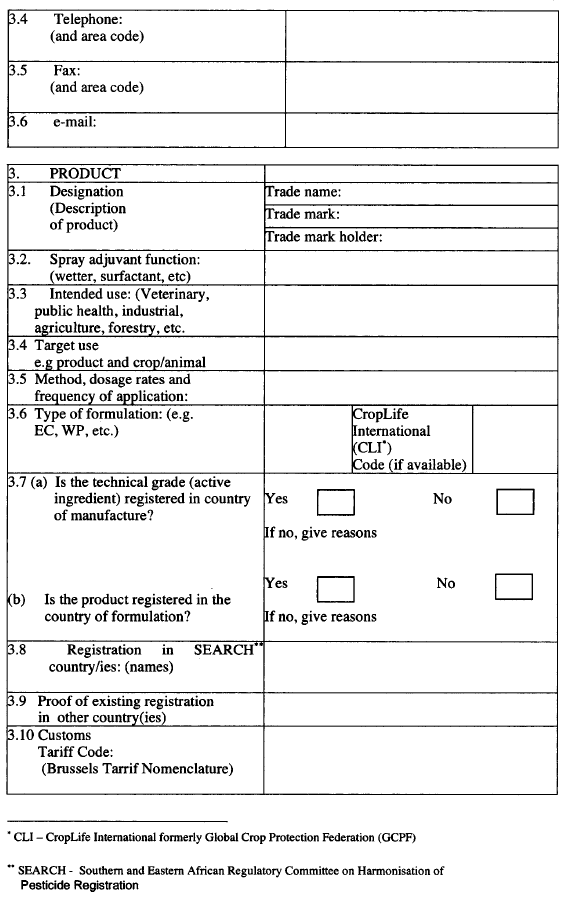
No label shall be used on a pest control product unless it has been approved by the Board and, unless the Board otherwise directs, every label shall show—
| (a) |
the name of a pest control product which shall be descriptive of the physical form and the purpose of the pest control product and shall include the common name of its active ingredients and may include a distinctive brand or trade mark;
|
| (b) |
the class designation of the pest control product in capital letters and shall be classified as set out in the First Schedule;
|
| (c) |
information detailing the nature and degree of hazard inherent in the pest control product and the nature and degree of hazard shall be identified by the appropriate precautionary symbol and signal words selected from the Second Schedule together with a statement respecting the nature of the primary hazard to which the symbol relates;
|
| (d) |
a statement directing the user to read the label which statement shall be in the following form – "READ THE LABEL BEFORE USING";
|
| (e) |
a guarantee statement in the following manner—
| (i) |
the word in capital letters "GUARANTEE", followed by; |
| (ii) |
a colon; followed by; |
| (iii) |
the common name of the active ingredient of pest control product or where a common name has not been designated, the chemical or other name of the active ingredient; followed by; |
| (iv) |
the contents of the active ingredient expressed— |
|
| (f) |
the registration number of the pest control product which shall be set out in the following manner—
"REGISTRATION NO. PCPB (CR) 0000" AND PCPB (TCR) 0000";
Where "CR" means Certificate of Registration for a period of three years and or renewal for a period not exceeding two years at any one time; and "TCR" means Temporary Certificate of Registration for a period not exceeding one year;
|
| (g) |
a statement of the net contents of the package for the pest control product, which shall be expressed—
| (i) |
in the case of a liquid pest control product of a volume less than one litre, in terms of millilitres; |
| (ii) |
in the case of a liquid pest control product of a volume of one litre or more, in terms of litres; |
| (iii) |
in the case of a pressure-packed pest control product in terms of weight, in grams if less than one kilogram; |
| (iv) |
in the case of a pressure-packed pest control product which weighs one kilogram or more, in terms of kilograms; |
| (v) |
in the case of a dry formulation pest control product which weighs less than a kilogram, in terms of grams; |
| (vi) |
in the case of a dry formulation pest control product which weighs one kilogram or more, in terms of kilograms; |
| (vii) |
in the case of a pest control product that is semi-solid or viscous in terms of either weight or volume in accordance with this paragraph; or |
| (viii) |
in the case of a pest control product the nature of which does not lend itself to net content statements prescribed in this paragraph, in terms acceptable to the Board; |
|
| (h) |
the name and postal address of the registrant and the name and postal address of the resident agent, if any and the names and postal addresses of manufacturer, formulator, distributor and re-packer;
|
| (i) |
the directions for use of the pest control product, which directions shall include dosage rates, timing of application and use limitations;
|
| (j) |
information identifying any significant hazards respecting the handling, storage, display, distribution and disposal of the pest control product which information shall include instructions respecting procedures to alleviate the hazard and when required by the Board, instructions respecting decontamination procedures and disposal of the pest control product and the empty package;
|
| (k) |
information identifying any significant hazard to—
| (i) |
things on or in relation to which the pest control product is intended to be used; or |
| (ii) |
public health, plants, animals or the environment, which information shall include instructions respecting the procedures to alleviate any such hazard; |
|
| (l) |
instructions for first aid, which instructions shall be under the heading in capital letters "FIRST AID INSTRUCTIONS" and shall set out the practical measures to be taken in the event of poisoning or other injury caused by the pest control product;
|
| (m) |
the toxicological information essential to the treatment of a person poisoned or otherwise injured by the pest control product, which information shall be under the heading in capital letters "TOXICOLOGICAL INFORMATION" and shall—
| (i) |
describe the symptoms of poisoning; |
| (ii) |
state antidotes and remedial measures; and |
| (iii) |
state the ingredient that may affect the treatment; |
|
| (n) |
a notice to the user of the pest control product which notice shall be in the following manner;
|
|
This pest control product is to be used only in accordance with the directions on this label. It is an offence under the Pest Control Products Act (Cap. 346) to use or store a pest control product under unsafe conditions;
| (o) |
the colour codes banding precautionary pictograms relating to the toxicity of the product in the following manner of declining toxicity—
| (i) |
PMS Red 199C for products classified under WHO class Ia and Ib; |
| (ii) |
PMS yellow C for products classified under WHO class II; |
| (iii) |
PMS Blue 293C for products classified under WHO class III; |
| (iv) |
PMS Green 347C for unclassified products under WHO classification; |
| (v) |
the colour shall not be used elsewhere on the label; and |
| (vi) |
any other pictogram as directed by the Board; |
|
| (p) |
separate pictograms depicting at least one pest controlled by the pest control product and crop or animal or other usage as registered under this Act;
|
| (q) |
an instruction directing the user on storage which shall be in capital letters– KEEP LOCKED OUT OF REACH OF CHILDREN;
|
| (r) |
information on the date of manufacture, expiry date, batch number and shelf-life if stored in original container under cool dry conditions.
|
| (3) |
The label for a pest control product which is a device of a type or kind listed in the First Schedule to the Pest Control Products (Registration) Regulations shall contain the information referred to in subparagraphs (f), (h), (i), (j), and (k) of paragraph (2).
[L.N. 127/2006, r. 3.]
|
|
| 4. |
Display panels
| (1) |
The display panel shall consist of one principal display panel and at least one secondary display panel.
|
| (2) |
Where the primary purpose of a pest control product is not to control, prevent, destroy, mitigate, attract or repel a pest, but it has these properties, the pest control product shall have a display panel with—
| (a) |
the information referred to in subparagraphs (a), (b), (c), (d), (g) and (h) of regulation 3(2) on the principal display panel; and
|
| (b) |
the information referred to in subparagraphs (e), (f), (i), (l) and (m) of regulation 3(2) on the secondary display panel.
|
|
| (3) |
Where the primary purpose of a pest control product is to control, prevent, destroy, mitigate, attract or repel a pest, the pest control product shall have a display panel with the information referred to in subparagraphs (a), (b), (c), (d), (e), (f), (g) and (h) of regulation 3(2) on the principal display panel.
|
| (4) |
This Regulation shall apply to a label for a pest control product that is within the meaning of paragraph (a) of the definition of "pest control product" in section 2 of the Act.
|
|
| 5. |
Notice on display panel
| (1) |
Where the principal display panel shows the pest control product as "RESTRICTED", the notice referred to in regulation 3(2)(n) shall appear prominently at the top of the secondary display panel followed by the heading in capital letters "RESTRICTED USE", followed by the directions for use, dosage, timing of application and use limitations to which the restrictions relates all of which shall be circumscribed by a line to set the information apart from all other information required to be shown on the secondary display panel.
|
| (2) |
Notwithstanding paragraph (1), where the principle display panel shows the pest control product as "RESTRICTED" the directions for use, dosage rates, timing of application and use limitations to which the restriction relates, together with the information referred to in subparagraphs (a), (b), (c), (d), (e), (f), (g), (h) and (i) of regulation 3(2) may, with the approval of the Board, appear in a brochure or leaflet that will accompany the package for the pest control product.
|
| (3) |
Where the information required to be shown on the label is not included in the display panel, the display panel shall contain the words in capital letters "READ ATTACHED BROCHURE BEFORE USE" prominently displayed thereon.
|
|
| 6. |
Display of additional information
| (1) |
Subject to the approval of the Board, additional information relating to the pest control product and any graphic design or symbol maybe shown on the label if it does not unreasonably detract or obscure the information required to be shown on the label under these Regulations.
|
| (2) |
A registrant may include on the label the following liability limitation warranty—
Seller’s guarantee is limited to the terms set out on the label and subject thereto, the buyer assumes the risk to persons or property arising from the use or handling of this product and accepts the product on that condition.
|
|
| 7. |
Information where product is distributed in bulk
Where a pest control product is distributed in a bulk container, the information referred to in subparagraphs (a), (b), (e), (f), (g), (h), (i) and (m) of regulation 3(2) shall be shown—
| (a) |
on the bulk container; and
|
| (b) |
on the documents respecting the pest control product or on a statement accompanying the consignment.
|
|
| 8. |
Information on label
| (1) |
The information on every label shall be printed in both English and Kiswahili languages.
|
| (2) |
All units of measures shown on the label shall be expressed in accordance with the requirements of the Weights and Measures Act (Cap. 513).
|
| (3) |
All information shown on the label shall be printed in a manner that is conspicuous, legible and indelible.
|
|
| 9. |
Denaturation
Where the physical properties of a pest control product are such that the presence of the pest control product may not be recognized when it is used, and is likely to expose a person or domestic animal to a severe health risk, the pest control product shall be denatured by means of colour, odour or such other means as the Board may approve to provide signal or warning of its presence.
|
| 10. |
Storage and display
A pest control product shall be stored and displayed in accordance with the conditions shown on the label.
|
| 11. |
Distribution
Notwithstanding the provisions of the Pharmacy and Poisons Act (Cap. 244), a pest control product shall be distributed in accordance with such condition as the Board shall specify.
|
| 12. |
Prohibition
No person shall use a pest control product in a manner that is inconsistent with the directions or limitations respecting its use as shown on the label.
|
| 13. |
Packaging
| (1) |
Every package for a pest control product shall, unless the Board otherwise directs, be approved by the Board.
|
| (2) |
The package for every pest control product shall be sufficiently durable and be designed and manufactured to contain the pest control product safety under practical conditions of storage, display and distribution.
|
| (3) |
Every package shall be designed and manufactured to permit—
| (a) |
the withdrawal of any or all of the contents in a manner that is safe to the user; and
|
| (b) |
the closing of the package in a manner that will contain the pest control product satisfactorily under practical conditions.
|
|
| (4) |
Every package shall be constructed in such a manner as to minimize the degradation or change of its contents resulting from interaction or from the effects of radiation or other means.
|
|
| 14. |
General prohibitions
| (1) |
A label shall not contain any information respecting any organism or causative agent of a disease that is required to be reported under the Animal Diseases Act (Cap. 364).
|
| (2) |
Information that is required under these Regulations to be shown on a label shall not appear at the bottom of the package.
|
| (3) |
Words stating, implying or inferring that a pest control product is approved, accepted or recommended by the government or by any department or agency thereof shall not appear on a package or label or in any advertisement respecting a pest control product.
|
|
FIRST SCHEDULE
[Regulation 3(2), L.N. 127/2006, ss. 4, 5 and 6.]
CLASSIFICATION OF PEST CONTROL PRODUCTS
| 1. |
Restricted Class
Where the Board has set forth additional essential conditions to be shown on the label respecting the display, distribution, use, limitations or qualifications of persons who may use the pest control product, the pest control product shall have the following properties—
| (a) |
acute oral LD50 is greater than 50 mg/kg body weight;
|
| (b) |
acute dermal LD50 is greater than 100 mg/kg body weight;
|
| (c) |
environmental risks are significant and will be judged accordingly;
|
| (d) |
pest control products used in aquatic and forestry situations are classified restricted.
|
[L.N. 127/2006, rr. 4 and 5.]
|
| 2. |
Commercial and Agricultural Class
Where the pest control product is to be displayed and distributed for general use in commercial activities specified on the label it shall have the following properties—
| (a) |
acute oral LD50 is greater than 50 mg/kg body weight;
|
| (b) |
acute dermal LD50 is greater than 100 mg/kg body weight;
|
| (c) |
environmental effects possible in limited regions.
|
|
| 3. |
Domestic Class
Where the pest control product is to be displayed and distributed for the use in and around a dwelling it shall have the following properties—
| (a) |
acute oral LD50 is over 500 mg/kg body weight;
|
| (b) |
acute dermal LD50 is over 1,000 mg/kg body weight;
|
| (c) |
no special precautions or equipment required for inhalation hazard;
|
| (d) |
no irreversible effects from repeated exposures;
|
| (e) |
disposal of product and containers can safely be done by placing in garbage;
|
| (f) |
package sizes limited to amounts that can be safely used and stored by consumers.
|
|
SECOND SCHEDULE
CAUTIONARY SYMBOLS AND WORDS
THE PEST CONTROL PRODUCTS (REGISTRATION) REGULATIONS, 1984
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Registration) Regulations, 1984.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
“accredited scientists/institution” means a person or an institution that has been officially recognized by the Board as having the capacity and competence to undertake biological efficacy trials;
“active ingredient” means an ingredient of a pest control product to which the effects of the pest control product are attributed and includes a synergist; but does not include a component that by itself is not primarily responsible for the control effect of the pest control product;
“biochemical pesticide” means a pest control product whose active ingredient constitutes a chemical derived from naturally occurring plant or animal intended to control invertebrate pests;
“certificate of registration” means a certificate issued by the Board under regulation 7;
“device” means any article, instrument, apparatus, contrivance or gadget that by itself or in conjunction with a pest control product is used as a means to control pests directly or indirectly;
“experimental permit” means a permit issued by the Board for small quantity of a pest control product imported or produced locally for purposes of research and efficacy trials prior to consideration for registration;
“microbial and macrobial biopesticide” means a pest control product of naturally occurring micro-organisms (microbiological agents viruses and rickettsia; bacteria, protozoa, fungi,) and macro-organisms (macro biological agents such as predators, parasitoids and entomopathogenic nematodes), respectively intended for the control of invertebrate pests weeds, pathogens of crops, and pests of livestock and public health and to which effects of the pest control products or active agent are attributed but does not include a component that by itself is not primarily responsible for the control effect of the pest control product or genetically modified living microorganism and macro-organism;
“national collection number” means the unique code given to a culture or an isolate by the National Museums of Kenya;
“parallel/daughter registration” means a registration of a trade name based on the strength of an existing fully registered product from the same manufacturer and source and with authorization from the registrant;
“residue” means the ingredient of a pest control product that remains after the pest control product has been used and includes substances resulting from degradation or metabolism.
[L.N. 123/2006, r. 2, L.N. 124/2015, r. 2.]
|
| 3. |
Exemption from registration
A pest control product shall be exempt from registration if—
| (a) |
it is for use by a person for research purposes if that use has been approved by the Board;
|
| (b) |
it is a type or kind set out in the First Schedule and meets the conditions relevant to that substance as set out in that Schedule.
|
|
| 3A. |
Efficacy testing
| (1) |
Every person desiring to introduce a pest control product for efficacy testing shall—
| (a) |
make application to the Board for an experimental permit in Form C set out in the Second Schedule;
|
| (b) |
provide all the details required in the form;
|
| (c) |
on request supply any further information which may be required by the Board; and
|
| (d) |
pay the prescribed application fees determined by the Board from time to time therefor.
|
|
| (2) |
| (a) |
The Board shall evaluate the data provided and if satisfied that the application merits approval, shall issue an experimental permit in the prescribed Form D in the Second Schedule
|
| (b) |
The Board shall, in addition, give the applicant information relating to the existing accredited scientists or institutions in the field of trial whom the applicant will work with;
|
|
| (3) |
When the efficacy trials are complete the accredited scientist or institution shall submit efficacy reports to the Board.
[L.N. 123/2006, r. 3.]
|
|
| 4. |
Application for registration of pest control products
| (1) |
Every person desiring to register a pest control product shall make application to the Board in either Forms A, A1, A2, A3, A3a, A3b, A4 or A5 set out in the Second Schedule and shall, on request, supply any further information which may be required by the Board.
|
| (2) |
An applicant who is not resident in Kenya shall appoint an agent permanently resident in Kenya to whom any notice or correspondence may be sent.
|
| (2A) |
An application for registration of an agent shall be submitted in Form A6 set out in the Second Schedule.
|
| (2B) |
An application for registration for change of an agency shall be submitted in Form A7 set out in the Second Schedule.
|
| (3) |
An application for the registration of a pest control product shall be accompanied by five copies of the proposed label for the pest control product or reasonable facsimiles thereof.
|
| (4) |
An applicant who is not resident in Kenya shall be required to deposit with the Board a binding agreement entered with the agent permanently resident in Kenya.
[L.N. 123/2006, r. 4, 5, 6, L.N. 124/2015, r. 3.]
|
|
| 4A. |
Application for registration of a synthetic or conventional pest control product
| (1) |
The application for registration of a synthetic or conventional pest control product (non-generic) under regulation 4 (1) shall be in the prescribed Form A set out in the Second Schedule completed by the applicant or duly authorized person and submitted in triplicate.
|
| (2) |
The application for registration of generic conventional pest control products shall be submitted in Form A4.
|
| (3) |
The Board shall supply the applicant with checklists and an index to ensure that the applicant has supplied the relevant data required in Form A set out in the Second Schedule.
[L.N. 124/2015, r. 4.]
|
|
| 4B. |
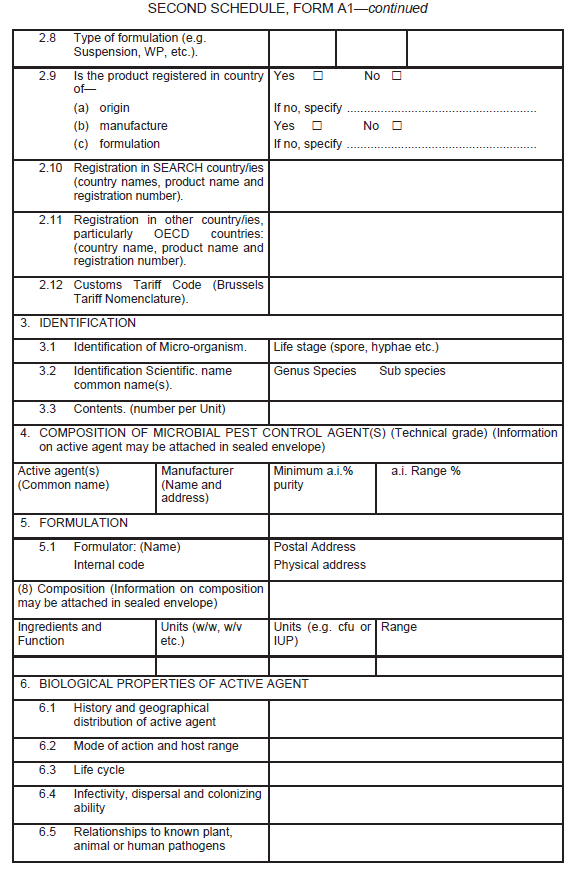
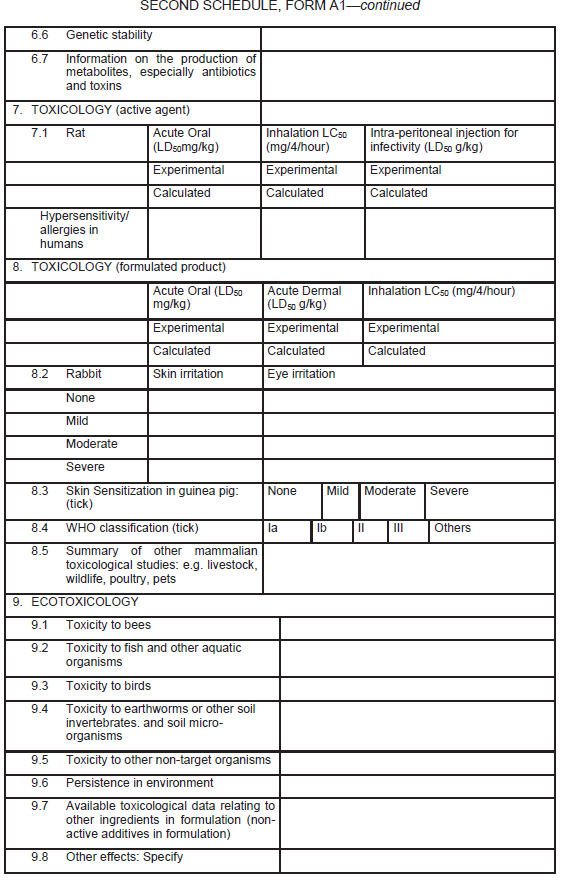
Application for the registration of microbial biopesticide
| (1) |
An application for the registration of microbial biopesticide shall be in Form A1 set out in the Second Schedule.
|
| (2) |
Information in support of a request for registration, both published and unpublished (fully cited), shall be supplied in the form of a summary data sheet laid out in the format given in Form A1 set out in the Second Schedule.
|
| (3) |
Pre-registration consultations between the applicant and the Board shall be undertaken after the application is made.
|
| (4) |
All applicants intending to import or export live organisms into or out of the country shall comply with all other existing laws governing such organisms.
|
| (5) |
The use of genetically modified organisms as microbial biopesticides shall comply with any other existing laws governing such organisms before an application is made to the Board.
|
| (6) |
The Board shall supply checklists and an index to ensure that the applicant has provided all relevant data and cited material.
[L.N. 124/2015, r. 5.]
|
|
| 4C. |
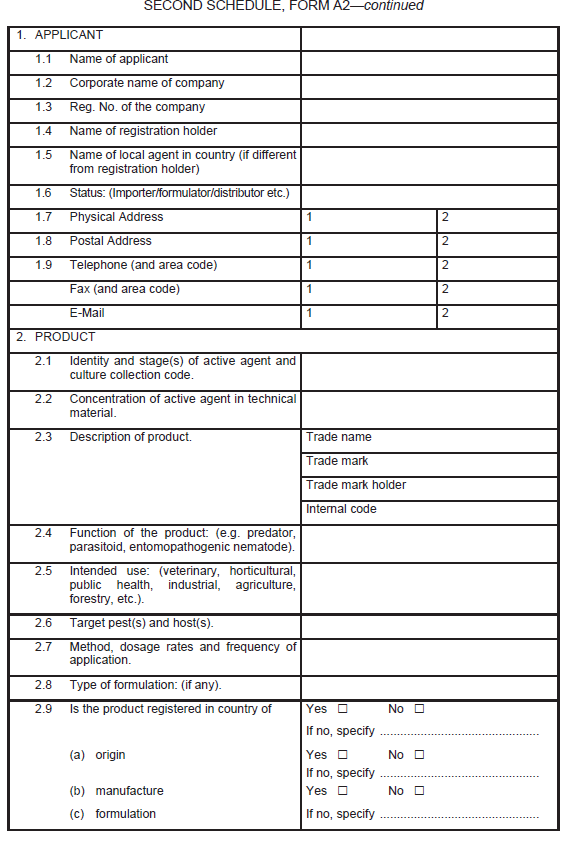
Application for the registration of macrobial biopesticide
| (1) |
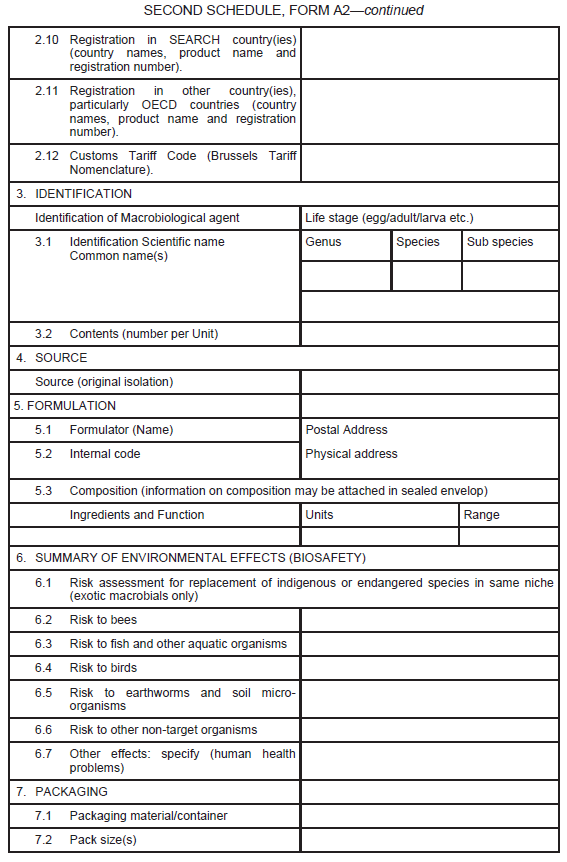
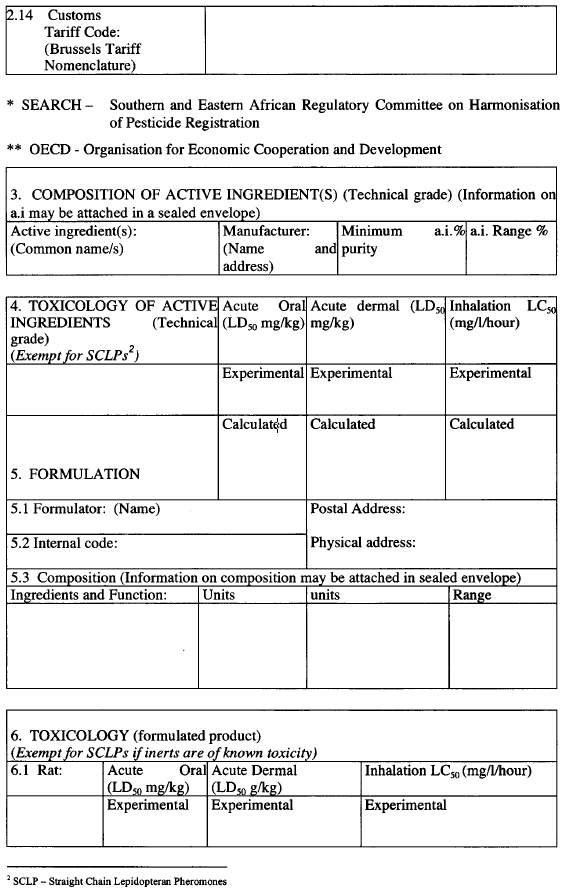
An application for the registration of macrobial biopesticide shall be as set out in Form A2 in the Second Schedule.
|
| (2) |
Information in support of a request for registration, both published and unpublished (fully cited), shall be supplied in the form of a summary data sheet laid out in the format given in Form A2 set out in the Second Schedule.
|
| (3) |
Pre-registration consultations between the applicant and the Board shall be undertaken after the application is made.
|
| (4) |
The applicant shall be required to-
| (a) |
submit a sample of the pest control product to the National Museums of Kenya or the national collection number obtained if the culture is already in collection;
|
| (b) |
provide a sample of the technical grade of its active agent;
|
| (c) |
send an additional sample to the National Agricultural Research Laboratories, Biological Control Unit, Kenya Agricultural Research Institute and Kenya Plant Health Inspectorate Service;
|
| (d) |
supply any other sample as may be requested by the Board.
|
|
| (5) |
All applicants intending to import or export live organisms into or out of the country shall comply with any other existing laws governing such organisms.
|
| (6) |
The use of genetically modified organisms and living modified organisms as macrobial biopesticides shall comply with any other existing laws governing such organisms before an application is made to the Board.
|
| (7) |
The Board shall supply checklists and an index to ensure that the applicant has provided all relevant data and cited material.
[L.N. 124/2015, r. 6.]
|
|
| 4D. |
Application form for the registration of a biochemical pesticide other than semiochemicals
| (1) |
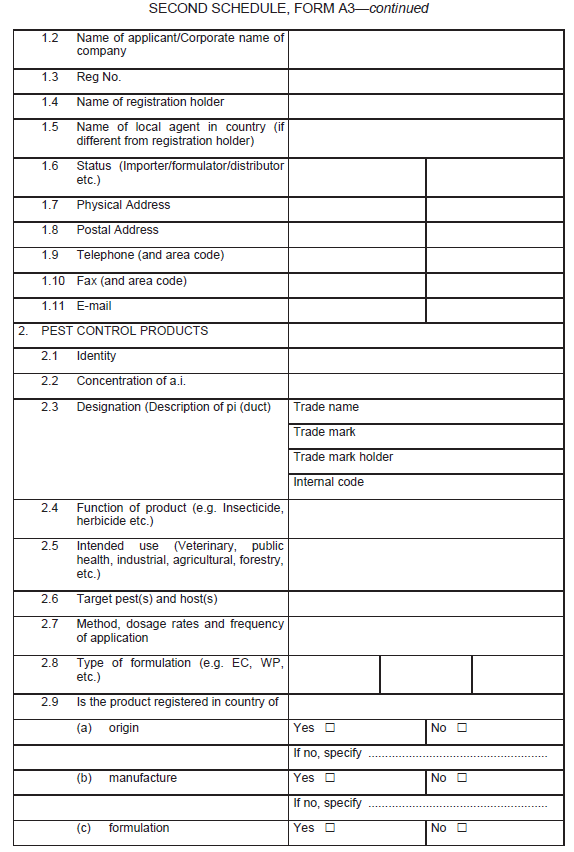
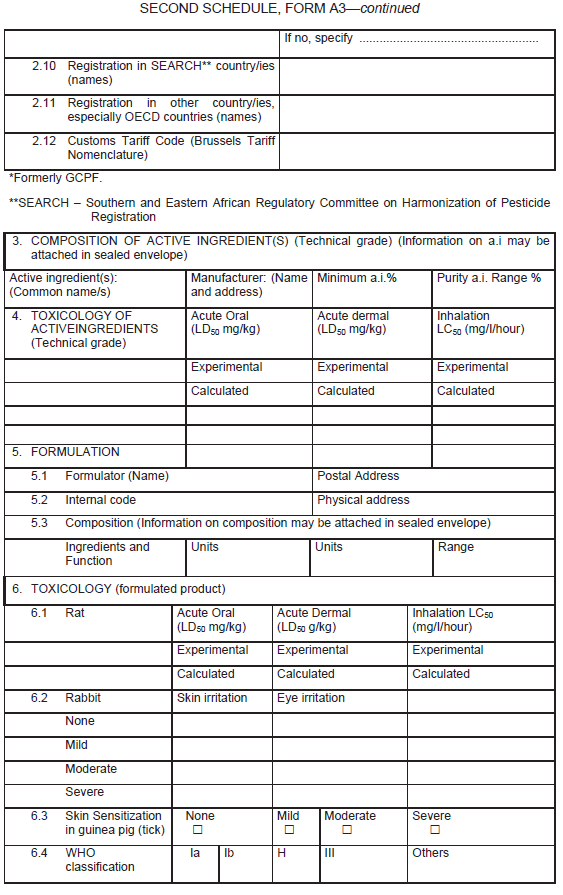
The application form for the registration of a biochemical pesticide other than semiochemicals, shall be in Form A3 in the Second Schedule.
|
| (2) |
Information in support of a request for registration both published and unpublished shall be supplied in form of a summary data sheet laid out according to the format given in Form A3.
|
| (3) |
Pre-registration consultation between the applicant and the registration authority shall be undertaken.
|
| (4) |
The application for registration of semiochemicals pest control products for monitoring and control of pests in crops, livestock and public health shall be in Form A3a.
|
| (5) |
The application for introduction of straight chained lepidopteran pheromone for monitoring purposes shall be in Form A3b.
[L.N. 124/2015, r. 7.]
|
|
| 4E. |
Application form for registration of spray adjuvants
| (1) |
The application form for registration of spray adjuvants shall be in Form A5 as set out in the Second Schedule.
|
| (2) |
Information in support of a request for registration, both published and unpublished (fully cited) shall be supplied in the form of a summary data sheet laid out in Form A5 in the Second Schedule.
|
| (3) |
The Board shall supply checklists and an index to ensure that the applicant has provided all relevant data and cited material.
[L.N. 124/2015, r. 8.]
|
|
| 4F. |
Registration of a parallel/daughter pest control product
| (1) |
The Board may upon such terms and conditions as it may specify, on payment of the introductory and registration fees, register a parallel/daughter pest control product where the applicant-
| (a) |
completes Form C set out in the Second Schedule;
|
| (b) |
provides a letter of access from registrant and a letter of no objection from the local agent;
|
| (c) |
submits a letter of no objection from the trade name owner where the trade name owner is not the registrant;
|
| (d) |
borrows approved label of the original registered product and only changes the trade name.
|
|
| (2) |
Each parallel/daughter registration shall have its own registration number which shall be linked to the original registered product by indicating the original registered product number on the parallel registration certificates.
|
| (3) |
Voluntary cancellation of a product shall apply to the registered product and the parallel/daughter products.
|
| (4) |
A company may leave an area without canceling the parallel/daughter registration and may transfer access of the original dossier thereto.
|
| (5) |
The parallel/daughter registration shall be automatically revoked when the registrant withdraws the letter of access.
|
| (6) |
The paralle/daughter registration shall not be used to register other different products.
|
| (7) |
A new dossier shall not be required for the registration of the parallel/daughter products;
|
| (8) |
The original registered product and the paralle/daughter registration shall be required to originate from the same source.
|
| (9) |
Parallel/daughters registrations shall be exempt from local efficacy trials if the intended use is identical to that of the original registered product.
|
| (10) |
Efficacy trials shall be undertaken where new uses which are different from those of the original registered products.
|
| (11) |
The Board shall exercise discretion in determining the number of parallel/daughter products to be registered on a case-by-case basis but not more than five such products shall be registered in respect of one original product.
[L.N. 124/2015, r. 8.]
|
|
| 4G. |
Extension of the use of a registered pest control product
The Board may, upon such terms and conditions as it may specify, extend the use of a registered pest control product through a label extension where the applicant submits-
| (a) |
successful two-season efficacy trial data on the respective area of use such as crop or pest combination;
|
| (b) |
residue data based on the Good Agriculture Practice in the efficacy trial, if the new use is on edible crops or animals;
|
| (c) |
a copy of the previously approved label;
|
| (d) |
revised commercial label with the proposed new uses, rates, pre-harvest interval and re-entry interval and the final commercial version label;
|
| (e) |
proposed maximum residue limits and pre-harvest intervals for edible commodities, withdrawal period for livestock and re-entry interval for greenhouse use.
|
|
| 4H. |
Summary dossier
The application for registration of pest control products shall be accompanied by a copy of a summary dossier as prescribed in form-
| (a) |
B for conventional pest control products;
|
| (b) |
B1 for microbial pest control products;
|
| (c) |
B2 for macrobial pest control products;
|
| (d) |
B3 for biochemical pest control products other than semiochemicals; and
|
|
| 5. |
Applicant to provide samples
| (1) |
An applicant shall, when requested to do so by the Board, provide—
| (a) |
a sample of the pest control product;
|
| (b) |
a sample of the technical grade of its active ingredient;
|
| (c) |
a sample of the laboratory standard of its active ingredient; and
|
| (d) |
any other sample as may be required by the Board.
|
|
| (2) |
The samples shall be submitted to the Board in Form A8 set out in the Second Schedule.
[L.N. 124/2015, r. 9.]
|
|
| 6. |
Registration fees
The fees payable by an applicant for the registration of a pest control product shall be the prescribed fees determined by the Board from time to time where a pest control product is a device or it contains an active ingredient that has been previously assessed or evaluated for the purpose of the Act and these Regulations.
|
| 7. |
Issue of certificate of registration
| (1) |
The Board shall consider the application under regulation 4, and, if it is satisfied of the safety, efficacy, quality and economic value of the pest control product, shall register the pest control product, and issue a certificate of registration which shall be in Form B set out in the Second Schedule.
|
| (1A) |
The applicant shall submit the proposed trade name for consideration by the Board.
|
| (1B) |
The trade name in sub-regulation (1A) may be changed upon request to the Board in Form A9 set out in the Second Schedule.
|
| (1C) |
The fees payable by an applicant for the change of a trade name of a pest control product shall be ten thousand shillings.
|
| (2) |
If the Board is not satisfied as to the safety, efficacy, quality and economic value of the pest control product it may, after providing an opportunity for the applicant to be heard, reject the application for the registration of the pest control product and inform the applicant the reasons for the rejection in writing.
|
| (3) |
No person to whom a certificate of registration has been issued under this Regulation shall lend, hire, sell, transfer or otherwise dispose of the certificate to any other person without the approval of the Board, which approval shall be endorsed on the certificate of registration.
[L.N. 124/2015, r. 10.]
|
| (1) |
Any person who intends to handle, use, distribute, transport or deal in a Pest Control Product under restricted class shall apply to the Board for a permit in the prescribed Form D in the Schedule.
|
| (2) |
No person shall handle, use, distribute, transport or deal in a Pest Control Product under restricted class without a permit from the Board in the prescribed Form E in the Schedule.
[LN 124 of 20006, s. 7.]
|
|
| 9. |
Temporary registration
| (1) |
The Board may upon such terms and conditions as it may specify, on payment of a prescribed fee determined by the Board from time to time, register a pest control product for a period not exceeding one year where—
|
| (2) |
Any terms and conditions specified by the Board under paragraph (1) shall be contained in the temporary certificate of registration.
|
|
| 10. |
Refusal to register pest control
The Board may refuse to register a pest control product if in its opinion—
| (a) |
the applicant for registration or the label for the pest control product does not comply with, the provisions of the Act and these Regulation;
|
| (b) |
the information provided to the Board by the applicant is insufficient to enable the pest control product to be assessed or evaluated;
|
| (c) |
the applicant fails to establish that the pest control product has merit or value for the purpose claimed when the pest control product is used in accordance with its label directions; or
|
| (d) |
the use of the pest control product would lead to an unacceptable risk or harm to—
| (i) |
things on or in relation to which the pest control product in intended to be used; or |
| (ii) |
public health, plants, animals or the environment. |
|
|
| 11. |
Suspension and revocation of certificates of registration
| (1) |
The Board may suspend, amend or revoke a certificate of registration issued under these Regulations for such time as the Board may determine.
|
| (2) |
The powers conferred by paragraph (1) shall not be exercised by the Board except on one or more of the following grounds—
| (a) |
that the matters stated in the application on which the certificate of registration was granted were false or incomplete in a material particular;
|
| (b) |
that new information has become available to the Board which renders the pest control product unsafe or dangerous;
|
| (c) |
that the premises on which, or on part of which, the pest control product is manufactured, assembled or stored by or on behalf of the holder of the certificate of registration are unsuitable for the manufacturing, assembly or storage of pest control products;
|
| (d) |
that the holder of a certificate of registration has given a notice to the Board in writing of any intentions to suspend product registration for a period not exceeding 5 years;
|
| (e) |
that new information has become available to the Board indicating that the pest control product is sourced from a manufacturer, formulator, repacker or any facility other than that specified in the application forms and dossier for registration for the respective pest control product or sources authorized by the Board;
|
| (f) |
that the principal or registrant withdraws the technical support to the local agent or distributor on the basis of which a pest control product was registered, in writing;
|
| (g) |
that the new information has become available to the Board which renders the pest control product ineffective, not efficacious or of no economic value for the purposes for which it is intended;
|
| (h) |
that a holder of a registered pest control product has in connection with the registration concerned, contravened or failed to comply with a provision of this Act;
|
| (i) |
that a holder of such registration has contravened or failed to comply with a condition to which the registration concerned is subject to;
|
| (j) |
that it is contrary to the public interest that such pest control product should remain registered;
|
| (k) |
that any incorrect or misleading advertisement is used in connection with any registered pest control product.
|
|
|
| 12. |
Notice to holder of certificate of registration, etc.
| (a) |
refuses to register a pest control products; or
|
| (b) |
suspends or revokes the certificate of registration, it shall send to the applicant or the holder of a certificate of registration, as the case may be, a notice by registered post notifying him of the refusal, suspension or revocation.
|
|
| 13. |
Appeals
| (1) |
An applicant or holder of a certificate of registration who has received a notice under regulation 12 may within thirty days from the date which the notice is received by him appeal to the Minister, who may amend or vary the decision as he thinks fit and whose decision shall be final.
|
|
| 14. |
Records
A holder of a certificate of registration issued under these Regulations shall keep a record of all the quantities of pest control products stored, manufactured or sold by him and the record shall—
| (a) |
be maintained for five years from the time it is made; and
|
| (b) |
be made available to the Board at such times and in much manner as the Board may require.
|
|
| 15. |
Declared source of product
| (1) |
Where a certificate of registration is issued under this regulation, the product shall be obtained from the declared source at the time of registration.
|
| (2) |
An application for change of source for the product shall be submitted to the Board as prescribed in Form E set out in the Second Schedule.
[L.N.124/2015, r. 11.]
|
|
FIRST SCHEDULE
ITEMS EXEMPTED FROM REGISTRATION
| 1. |
Garment bags, cabinets or chests that are manufactured, represented or sold as a means to protect clothing or fabrics from pests.
|
| 2. |
Electronic apparatus that is manufactured, represented or sold as a means to attract or destroy flying insects.
|
| 3. |
Devices of products that are manufactured, represented or sold to repel birds and other pests by causing physical discomfort by means of sound or touch
|
| 4. |
Devices of attachment to garden watering hoses that are manufactured, represented or sold as pest control product.
|
| 5. |
Devices that are manufactured, represented or sold as a means of providing the automatic or unattended application of a pest control product.
|
| 6 |
Devices that are sold for use with chemical products containing cyanide as a means to control animal pests.
|
THE PEST CONTROL PRODUCTS (REGISTRATION) REGULATIONS
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Registration) Regulations.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
“accredited scientists/institution” means a person or an institution that has been officially recognized by the Board as having the capacity and competence to undertake biological efficacy trials;
“active ingredient” means an ingredient of a pest control product to which the effects of the pest control product are attributed and includes a synergist; but does not include a component that by itself is not primarily responsible for the control effect of the pest control product;
“biochemical pesticide” means a pest control product whose active ingredient constitutes a chemical derived from naturally occurring plant or animal intended to control invertebrate pests;
“certificate of registration” means a certificate issued by the Board under regulation 7;
“device” means any article, instrument, apparatus, contrivance or gadget that by itself or in conjunction with a pest control product is used as a means to control pests directly or indirectly;
“experimental permit” means a permit issued by the Board for small quantity of a pest control product imported or produced locally for purposes of research and efficacy trials prior to consideration for registration;
“microbial and macrobial biopesticide” means a pest control product of naturally occurring micro-organisms (microbiological agents viruses and rickettsia; bacteria, protozoa, fungi,) and macro-organisms (macro biological agents such as predators, parasitoids and entomopathogenic nematodes), respectively intended for the control of invertebrate pests weeds, pathogens of crops, and pests of livestock and public health and to which effects of the pest control products or active agent are attributed but does not include a component that by itself is not primarily responsible for the control effect of the pest control product or genetically modified living microorganism and macro-organism;
“national collection number” means the unique code given to a culture or an isolate by the National Museums of Kenya;
“parallel/daughter registration” means a registration of a trade name based on the strength of an existing fully registered product from the same manufacturer and source and with authorization from the registrant;
“residue” means the ingredient of a pest control product that remains after the pest control product has been used and includes substances resulting from degradation or metabolism.
[L.N. 123/2006, r. 2, L.N. 124/2015, r. 2.]
|
| 3. |
Exemption from registration
A pest control product shall be exempt from registration if—
| (a) |
it is for use by a person for research purposes if that use has been approved by the Board;
|
| (b) |
it is a type or kind set out in the First Schedule and meets the conditions relevant to that substance as set out in that Schedule.
|
|
| 3A. |
Efficacy testing
| (1) |
Every person desiring to introduce a pest control product for efficacy testing shall—
| (a) |
make application to the Board for an experimental permit in Form C set out in the Second Schedule;
|
| (b) |
provide all the details required in the form;
|
| (c) |
on request supply any further information which may be required by the Board; and
|
| (d) |
pay the prescribed application fees determined by the Board from time to time therefor.
|
|
| (2) |
| (a) |
The Board shall evaluate the data provided and if satisfied that the application merits approval, shall issue an experimental permit in the prescribed Form D in the Second Schedule
|
| (b) |
The Board shall, in addition, give the applicant information relating to the existing accredited scientists or institutions in the field of trial whom the applicant will work with;
|
|
| (3) |
When the efficacy trials are complete the accredited scientist or institution shall submit efficacy reports to the Board.
[L.N. 123/2006, r. 3.]
|
|
| 4. |
Application for registration of pest control products
| (1) |
Every person desiring to register a pest control product shall make application to the Board in either Forms A, A1, A2, A3, A3a, A3b, A4 or A5 set out in the Second Schedule and shall, on request, supply any further information which may be required by the Board.
|
| (2) |
An applicant who is not resident in Kenya shall appoint an agent permanently resident in Kenya to whom any notice or correspondence may be sent.
|
| (2A) |
An application for registration of an agent shall be submitted in Form A6 set out in the Second Schedule.
|
| (2B) |
An application for registration for change of an agency shall be submitted in Form A7 set out in the Second Schedule.
|
| (3) |
An application for the registration of a pest control product shall be accompanied by five copies of the proposed label for the pest control product or reasonable facsimiles thereof.
|
| (4) |
An applicant who is not resident in Kenya shall be required to deposit with the Board a binding agreement entered with the agent permanently resident in Kenya.
[L.N. 123/2006, rr. 4, 5 & 6, L.N. 124/2015, r. 3.]
|
|
| 4A. |
Application for registration of a synthetic or conventional pest control product
| (1) |
The application for registration of a synthetic or conventional pest control product (non-generic) under regulation 4 (1) shall be in the prescribed Form A set out in the Second Schedule completed by the applicant or duly authorized person and submitted in triplicate.
|
| (2) |
The application for registration of generic conventional pest control products shall be submitted in Form A4.
|
| (3) |
The Board shall supply the applicant with checklists and an index to ensure that the applicant has supplied the relevant data required in Form A set out in the Second Schedule.
[L.N. 124/2015, r. 4.]
|
|
| 4B. |
Application for the registration of microbial biopesticide
| (1) |
An application for the registration of microbial biopesticide shall be in Form A1 set out in the Second Schedule.
|
| (2) |
Information in support of a request for registration, both published and unpublished (fully cited), shall be supplied in the form of a summary data sheet laid out in the format given in Form A1 set out in the Second Schedule.
|
| (3) |
Pre-registration consultations between the applicant and the Board shall be undertaken after the application is made.
|
| (4) |
All applicants intending to import or export live organisms into or out of the country shall comply with all other existing laws governing such organisms.
|
| (5) |
The use of genetically modified organisms as microbial biopesticides shall comply with any other existing laws governing such organisms before an application is made to the Board.
|
| (6) |
The Board shall supply checklists and an index to ensure that the applicant has provided all relevant data and cited material.
[L.N. 124/2015, r. 5.]
|
|
| 4C. |
Application for the registration of macrobial biopesticide
| (1) |
An application for the registration of macrobial biopesticide shall be as set out in Form A2 in the Second Schedule.
|
| (2) |
Information in support of a request for registration, both published and unpublished (fully cited), shall be supplied in the form of a summary data sheet laid out in the format given in Form A2 set out in the Second Schedule.
|
| (3) |
Pre-registration consultations between the applicant and the Board shall be undertaken after the application is made.
|
| (4) |
The applicant shall be required to-
| (a) |
submit a sample of the pest control product to the National Museums of Kenya or the national collection number obtained if the culture is already in collection;
|
| (b) |
provide a sample of the technical grade of its active agent;
|
| (c) |
send an additional sample to the National Agricultural Research Laboratories, Biological Control Unit, Kenya Agricultural Research Institute and Kenya Plant Health Inspectorate Service;
|
| (d) |
supply any other sample as may be requested by the Board.
|
|
| (5) |
All applicants intending to import or export live organisms into or out of the country shall comply with any other existing laws governing such organisms.
|
| (6) |
The use of genetically modified organisms and living modified organisms as macrobial biopesticides shall comply with any other existing laws governing such organisms before an application is made to the Board.
|
| (7) |
The Board shall supply checklists and an index to ensure that the applicant has provided all relevant data and cited material.
[L.N. 124/2015, r. 6.]
|
|
| 4D. |
Application form for the registration of a biochemical pesticide other than semiochemicals
| (1) |
The application form for the registration of a biochemical pesticide other than semiochemicals, shall be in Form A3 in the Second Schedule.
|
| (2) |
Information in support of a request for registration both published and unpublished shall be supplied in form of a summary data sheet laid out according to the format given in Form A3.
|
| (3) |
Pre-registration consultation between the applicant and the registration authority shall be undertaken.
|
| (4) |
The application for registration of semiochemicals pest control products for monitoring and control of pests in crops, livestock and public health shall be in Form A3a.
|
| (5) |
The application for introduction of straight chained lepidopteran pheromone for monitoring purposes shall be in Form A3b.
[L.N. 124/2015, r. 7.]
|
|
| 4E. |
Application form for registration of spray adjuvants
| (1) |
The application form for registration of spray adjuvants shall be in Form A5 as set out in the Second Schedule.
|
| (2) |
Information in support of a request for registration, both published and unpublished (fully cited) shall be supplied in the form of a summary data sheet laid out in Form A5 in the Second Schedule.
|
| (3) |
The Board shall supply checklists and an index to ensure that the applicant has provided all relevant data and cited material.
[L.N. 124/2015, r. 8.]
|
|
| 4F. |
Registration of a parallel/daughter pest control product
| (1) |
The Board may upon such terms and conditions as it may specify, on payment of the introductory and registration fees, register a parallel/daughter pest control product where the applicant-
| (a) |
completes Form C set out in the Second Schedule;
|
| (b) |
provides a letter of access from registrant and a letter of no objection from the local agent;
|
| (c) |
submits a letter of no objection from the trade name owner where the trade name owner is not the registrant;
|
| (d) |
borrows approved label of the original registered product and only changes the trade name.
|
|
| (2) |
Each parallel/daughter registration shall have its own registration number which shall be linked to the original registered product by indicating the original registered product number on the parallel registration certificates.
|
| (3) |
Voluntary cancellation of a product shall apply to the registered product and the parallel/daughter products.
|
| (4) |
A company may leave an area without canceling the parallel/daughter registration and may transfer access of the original dossier thereto.
|
| (5) |
The parallel/daughter registration shall be automatically revoked when the registrant withdraws the letter of access.
|
| (6) |
The paralle/daughter registration shall not be used to register other different products.
|
| (7) |
A new dossier shall not be required for the registration of the parallel/daughter products;
|
| (8) |
The original registered product and the paralle/daughter registration shall be required to originate from the same source.
|
| (9) |
Parallel/daughters registrations shall be exempt from local efficacy trials if the intended use is identical to that of the original registered product.
|
| (10) |
Efficacy trials shall be undertaken where new uses which are different from those of the original registered products.
|
| (11) |
The Board shall exercise discretion in determining the number of parallel/daughter products to be registered on a case-by-case basis but not more than five such products shall be registered in respect of one original product.
[L.N. 124/2015, r. 8.]
|
|
| 4G. |
Extension of the use of a registered pest control product
The Board may, upon such terms and conditions as it may specify, extend the use of a registered pest control product through a label extension where the applicant submits-
| (a) |
successful two-season efficacy trial data on the respective area of use such as crop or pest combination;
|
| (b) |
residue data based on the Good Agriculture Practice in the efficacy trial, if the new use is on edible crops or animals;
|
| (c) |
a copy of the previously approved label;
|
| (d) |
revised commercial label with the proposed new uses, rates, pre-harvest interval and re-entry interval and the final commercial version label;
|
| (e) |
proposed maximum residue limits and pre-harvest intervals for edible commodities, withdrawal period for livestock and re-entry interval for greenhouse use.
|
|
| 4H. |
Summary dossier
The application for registration of pest control products shall be accompanied by a copy of a summary dossier as prescribed in form-
| (a) |
B for conventional pest control products;
|
| (b) |
B1 for microbial pest control products;
|
| (c) |
B2 for macrobial pest control products;
|
| (d) |
B3 for biochemical pest control products other than semiochemicals; and
|
|
| 5. |
Applicant to provide samples
| (1) |
An applicant shall, when requested to do so by the Board, provide—
| (a) |
a sample of the pest control product;
|
| (b) |
a sample of the technical grade of its active ingredient;
|
| (c) |
a sample of the laboratory standard of its active ingredient; and
|
| (d) |
any other sample as may be required by the Board.
|
|
| (2) |
The samples shall be submitted to the Board in Form A8 set out in the Second Schedule.
[L.N. 124/2015, r. 9.]
|
|
| 6. |
Registration fees
The fees payable by an applicant for the registration of a pest control product shall be the prescribed fees determined by the Board from time to time where a pest control product is a device or it contains an active ingredient that has been previously assessed or evaluated for the purpose of the Act and these Regulations.
[L.N. 123/2006, r. 7.]
|
| 7. |
Issue of certificate of registration
| (1) |
The Board shall consider the application under regulation 4, and, if it is satisfied of the safety, efficacy, quality and economic value of the pest control product, shall register the pest control product, and issue a certificate of registration which shall be in Form B set out in the Second Schedule.
|
| (1A) |
The applicant shall submit the proposed trade name for consideration by the Board.
|
| (1B) |
The trade name in sub-regulation (1A) may be changed upon request to the Board in Form A9 set out in the Second Schedule.
|
| (1C) |
The fees payable by an applicant for the change of a trade name of a pest control product shall be ten thousand shillings.
|
| (2) |
If the Board is not satisfied as to the safety, efficacy, quality and economic value of the pest control product it may, after providing an opportunity for the applicant to be heard, reject the application for the registration of the pest control product and inform the applicant the reasons for the rejection in writing.
|
| (3) |
No person to whom a certificate of registration has been issued under this Regulation shall lend, hire, sell, transfer or otherwise dispose of the certificate to any other person without the approval of the Board, which approval shall be endorsed on the certificate of registration.
[L.N. 124/2015, r. 10.]
|
|
| 8. |
Duration and renewal of certificate of registration
| (1) |
A certificate of registration issued under these Regulations shall, unless earlier suspended or revoked, be valid for a period of three years from the date of issue and may thereafter be renewed for periods not exceeding two years at any one time.
|
| (2) |
The fee for the renewal of a certificate of registration shall be the prescribed fees determined by the Board from time to time, and an application for renewal shall be accompanied by five copies of the current label for the pest control product.
|
| (3) |
A holder of a certificate of registration issued under these Regulations shall give a notice to the Board in writing at least 3 months before the expiry of the registration of any intentions to keep the product registration in abeyance for a period not exceeding 5 years and the notice shall—
| (a) |
give reasons for temporary withdrawal; and
|
| (b) |
show the records of all quantities of the pest control product in stock, manufactured or sold by him.
|
|
| (4) |
The Board shall consider the notification under subregulation (3) and if it is satisfied with the reasons for temporary withdrawal shall suspend the registration of the pest control product for a period not exceeding 5 years.
|
| (5) |
The information on suspended registration under subregulation (4) shall be made known to the holder in writing and the general public by Gazette notice.
|
| (6) |
A person whose certificate of registration has been suspended under subregulation (4) shall withdraw the product from the market within a period of 3 months from the date of expiry of registration.
|
| (7) |
A person whose certificate of registration has been suspended under these Regulations shall give a notice to the Board in writing of any intentions to reintroduce the product registration and the notice shall—
| (a) |
give reasons for reintroduction,
|
| (b) |
be accompanied by a fee for the renewal of a certificate of registration for the preceding two years and the current year,
|
| (c) |
be accompanied by five copies of the current label for the pest control product.
|
|
| (8) |
A holder of a certificate of registration issued under these Regulations whose product registration has been suspended for a period exceeding five years shall apply for registration afresh and shall, on request supply any further information, which may be required by the Board.
[L.N. 123/2006, rr. 8 & 9.]
|
|
| 9. |
Temporary registration
| (1) |
The Board may upon such terms and conditions as it may specify, on payment of a prescribed fee determined by the Board from time to time, register a pest control product for a period not exceeding one year where—
|
| (2) |
Any terms and conditions specified by the Board under paragraph (1) shall be contained in the temporary certificate of registration.
[L.N. 123/2006, r. 10.]
|
|
| 10. |
Refusal to register pest control
The Board may refuse to register a pest control product if in its opinion—
| (a) |
the applicant for registration or the label for the pest control product does not comply with, the provisions of the Act and these Regulation;
|
| (b) |
the information provided to the Board by the applicant is insufficient to enable the pest control product to be assessed or evaluated;
|
| (c) |
the applicant fails to establish that the pest control product has merit or value for the purpose claimed when the pest control product is used in accordance with its label directions; or
|
| (d) |
the use of the pest control product would lead to an unacceptable risk or harm to—
| (i) |
things on or in relation to which the pest control product is intended to be used; or |
| (ii) |
public health, plants, animals or the environment. |
|
|
| 11. |
Suspension and revocation of certificates of registration
| (1) |
The Board may suspend, amend or revoke a certificate of registration issued under these Regulations for such time as the Board may determine.
|
| (2) |
The powers conferred by paragraph (1) shall not be exercised by the Board except on one or more of the following grounds—
| (a) |
that the matters stated in the application on which the certificate of registration was granted were false or incomplete in a material particular;
|
| (b) |
that new information has become available to the Board which renders the pest control product unsafe or dangerous;
|
| (c) |
that the premises on which, or on part of which, the pest control product is manufactured, assembled or stored by or on behalf of the holder of the certificate of registration are unsuitable for the manufacturing, assembly or storage of pest control products;
|
| (d) |
that the holder of a certificate of registration has given a notice to the Board in writing of any intentions to suspend product registration for a period not exceeding 5 years;
|
| (e) |
that new information has become available to the Board indicating that the pest control product is sourced from a manufacturer, formulator, repacker or any facility other than that specified in the application forms and dossier for registration for the respective pest control product or sources authorized by the Board;
|
| (f) |
that the principal or registrant withdraws the technical support to the local agent or distributor on the basis of which a pest control product was registered, in writing;
|
| (g) |
that the new information has become available to the Board which renders the pest control product ineffective, not efficacious or of no economic value for the purposes for which it is intended;
|
| (h) |
that a holder of a registered pest control product has in connection with the registration concerned, contravened or failed to comply with a provision of this Act;
|
| (i) |
that a holder of such registration has contravened or failed to comply with a condition to which the registration concerned is subject to;
|
| (j) |
that it is contrary to the public interest that such pest control product should remain registered;
|
| (k) |
that any incorrect or misleading advertisement is used in connection with any registered pest control product.
|
[L.N. 123/2006, r. 11, L.N.122/2014, r. 2.]
|
|
| 12. |
Notice to holder of certificate of registration, etc.
| (a) |
refuses to register a pest control products; or
|
| (b) |
suspends or revokes the certificate of registration,
|
it shall send to the applicant or the holder of a certificate of registration, as the case may be, a notice by registered post notifying him of the refusal, suspension or revocation.
|
| 13. |
Appeals
An applicant or holder of a certificate of registration who has received a notice under regulation 12 may within thirty days from the date which the notice is received by him appeal to the Minister, who may amend or vary the decision as he thinks fit and whose decision shall be final.
|
| 14. |
Records
A holder of a certificate of registration issued under these Regulations shall keep a record of all the quantities of pest control products stored, manufactured or sold by him and the record shall—
| (a) |
be maintained for five years from the time it is made; and
|
| (b) |
be made available to the Board at such times and in such manner as the Board may require.
|
|
| 15. |
Declared source of product
| (1) |
Where a certificate of registration is issued under this regulation, the product shall be obtained from the declared source at the time of registration.
|
| (2) |
An application for change of source for the product shall be submitted to the Board as prescribed in Form E set out in the Second Schedule.
[L.N. 124/2015, r. 11.]
|
|
FIRST SCHEDULE [r. 3]
ITEMS EXEMPTED FROM REGISTRATION
| 1. |
Garment bags, cabinets or chests that are manufactured, represented or sold as a means to protect clothing or fabrics from pests.
|
| 2. |
Electronic apparatus that is manufactured, represented or sold as a means to attract or destroy flying insects.
|
| 3. |
Devices of products that are manufactured, represented or sold to repel birds and other pests by causing physical discomfort by means of sound or touch.
|
| 4. |
Devices of attachment to garden watering hoses that are manufactured, represented or sold as pest control product.
|
| 5. |
Devices that are manufactured, represented or sold as a means of providing the automatic or unattended application of a pest control product.
|
| 6 |
Devices that are sold for use with chemical products containing cyanide as a means to control animal pests.
|
SECOND SCHEDULE [r. 4]
FORMS
[L.N. 123/2006.]
|
FORM A
|
(r. 4A(3))
|
|
APPLICATION FOR REGISTRATION OF A PEST CONTROL PRODUCT (CONVENTIONAL)
|
|
TRADE NAME OF THE PRODUCT .........................................................................................
|
|
PURPOSE OF APPLICATION (tick as appropriate) .............................................................
|
|
(a) Pest control product containing a new active ingredient ☐
|
|
(b) Pest control product where source of active and/or formulationis not identical to that of a registered product
|
☐
|
|
(c) Registration transfer
|
☐
|
|
(d) Amendments to existing registration
|
☐
|
|
(e) Other (Explain) .............................................................
|
|
Will the product he marketed under own label?
|
Yes ☐ No ☐
|
|
If no, Specify ...........................................................................
|
|
Proposed date of marketing ...............................................
|
|
1. APPLICANT
|
|
1.1 Identification
|
|
|
Name of applicant/Corporate name of company
|
|
|
Business Reg No.
|
|
|
Name of registration holder
|
|
|
Name of local agent in country:(If different from registration holder)
|
|
|
1.2 Status(Importer/formulator/distributor
|
|
|
Business Registration No.
|
|
|
1.3 Physical Address
|
|
|
1.4 Postal Address
|
|
|
1.5 Telephone: (and area code)
|
|
|
1.6 Fax: (and area code)
|
|
|
1.7 e-mail
|
|
|
2. PRODUCT
|
|
2.1 Designation (Description of product)
|
Trade name:
|
|
Trade mark:
|
|
Trade mark holder:
|
|
2.2 Function of product: (eg. Insecticide, herbicide etc.)
|
|
|
2.3 Intended use: (Veterinary, public health, industrial,agriculture, forestry, etc.)
|
|
|
|
|
|
2.4 Target pest(s) and host(s)
|
|
|
2.5 Method, dosage rates and frequency of application:
|
|
|
2.6 Types of formulation (e.g. EC, WP, etc.)
|
|
Crop life International (CLI*) Code (if available)
|
|
2.7 (a) Is the product registered in country of manufacture?(b) Is the product registered in the country of formulation?
|
Yes ☐ No ☐If no, give reasons
|
|
Yes ☐ No ☐If no, give reasons
|
|
2.8 Registration in SEARCH* country (ies) (names)
|
|
|
2.9 Existing registration No(s) and country (s)
|
|
|
2.10. Customs Tariff Code: (Brussels Tariff Nomenclature)
|
|
|
3. COMPOSITION OF ACTIVE INGREDIENT(S) (Technical grade) (information on a.i. may be attached in sealed envelope)
|
|
Active ingredients(s)(Common name(s)
|
Manufacture(Name and address)
|
Minimum a.i.%
|
purity| a.i. Range %
|
|
4. FORMULATION
|
|
4.1 Formulator: (Name)Postal AddressPhysical address
|
|
4.2 Internal code
|
|
4.3 Composition (Information on composition may be attached in sealed envelope)
|
|
Ingredients and Function
|
g/l
|
g/kg
|
Range
|
*Formerly GCPF
*SEARCH- Southern and Eastern African Regulation Committee on Harmonization of Pesticide Registration.
|
5.TOXICOLOGY (formulated product)
|
|
5.1 Rat
|
Acute Oral(LD50mg/kg)
|
Acute Denmal(LD50 mg/kg)
|
InhalationLC50 (mg/1/hour)
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
|
Calculated
|
Calculated
|
Calculated
|
|
5.2 Rabbit
|
Skin Irritation
|
Eye irritation
|
|
None
|
|
|
|
Mild
|
|
|
|
Moderate
|
|
|
|
Severe
|
|
|
|
5.3 Skin Sensitization in guinea pig: (tick)
|
None ☐ Mild ☐ Moderate ☐ Severe ☐
|
|
5.4 WHO classification
|
la
|
lb
|
II
|
III
|
Others
|
|
5.5. Summary of other mammalian toxicological studies : eg. livestock, wildlife, poultry, pets
|
|
5.6. Summary of environmental effects
|
|
5.6.1 Toxicity to bees
|
|
|
5.6.2 Toxicity to fish and other aquatic organisms
|
|
|
5.6.3 Toxicity to birds
|
|
|
5.6.4 Toxicity to earthworms and soil micro-organisms
|
|
|
5.6.5 Toxicity to other non-target organisms
|
|
|
5.6.6 Persistence in environment
|
|
|
5.6.7 Other effects: Specify:
|
|
|
6. PACKAGING
|
|
6.1 Packaging material/ container
|
|
|
6.2 Pack size(s)
|
|
|
6.3 Disposal of empty container(s)
|
|
7. OTHER SPECIFIC REQUIREMENTS
|
|
7.1 Human exposure
|
|
(a) Dermal absorption.(b) Likely human exposure under field conditions.(c) Available toxicological data relating to other ingredients in formulation (non-active additives in formulation).
|
|
|
8. DECLARATION
|
|
For and on behalf of ............................................I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete.
|
|
.............................................Name in full (printed)
|
......................................Signature
|
|
.......................................Official Title
|
....................................Date
|
|
Official Stamp of Applicant/Company
|
FOR OFFICIAL USERemarks..........................................................................................................................................................................
|
|
|
........................................Signed
|
...........................................Date
|
|
NOTE: The format of this application is recognized by all SEARCH countries.
|
_____________________________
|
FORM A1
|
(r. 4B(2))
|
|
APPLICATION FOR REGISTRATION OF A MICROBIAL BIOPESTICIDE (PEST CONTROL PRODUCTS-MICROBIAL AGENT)
|
|
TRADE NAME OF THE PRODUCT .....................................
|
|
PURPOSE OF APPLICATION (tick as appropriate) ......................
|
|
(a) Biopesticides containing a new active agent
|
|
(b) Biopesticides where source of active and/or formulation is not identical to that of a registered product
|
|
(c) Registration transfer
|
|
(d) Amendments to existing registration
|
|
(e) Other (Explain)
|
|
Will the product be marketed under own label? Yes ☐ No ☐If No, specify..................................
|
|
1. APPLICANT
|
|
Name of applicant
|
|
|
Corporate name of company
|
|
|
Reg No.
|
|
|
Name of registration holder:
|
|
|
Name of local agent in country: (If different from registration holder)
|
|
|
Status: (Importer/formulator/distributor etc.)
|
|
|
|
Physical Address
|
|
|
|
Postal Address
|
|
|
|
Telephone (and area code)
|
|
|
|
Fax (and area code)
|
|
|
|
E-mail
|
|
|
|
2. PRODUCT
|
|
2.1 Identity and stage (s) of active agent and culture collection code.
|
|
|
2.2 Concentration of active agent in technical material.
|
|
|
2.3 Designation (Description of product).
|
Trade name
|
|
Trade mark
|
|
Trade mark holder
|
|
Internal code
|
|
2.4 Function of product (e.g. Insecticide, herbicide etc).
|
|
|
2.5 Intended use: (Veterinary, horticultural, public health, industrial, agriculture, forestry, etc).
|
|
|
2.6 Target pests (s) and host(s).
|
|
|
2.7 Method, dosage rates and frequency of application.
|
|
|
2.8 Types of formulation (e.g. Suspension, WP, etc.).
|
|
|
|
|
2.9 Is the product registered in country of-(a) origin(b) manufacture(c) formation
|
Yes ☐ No ☐If No, specify ......................................................Yes ☐ No ☐If No, specify .............................................................
|
|
2.10 Registration in SEARCH country/ies (country name, product name and registration number).
|
|
|
2.11 Registration in other country/ies, particulary OECD countries: (country name, product name and registration number).
|
|
|
2.12 Custom Tariff Code (Brusells Tariff Nomenclature).
|
|
|
3. IDENTIFICATION
|
|
3.1 Identification of Micro-organism
|
Life stage (spore, hyphae etc.)
|
|
3.2 IdentificationScientific nameCommon name(s)
|
Genus
|
Species
|
Sub species
|
|
3.3 Contents (number per unit)
|
|
|
4. COMPOSITION OF MICROBIAL PEST CONTROL AGENT(S) (Technical grade) (Information on active agent may be attached in sealed envelope)
|
|
Active agent(s)(Common name/s)
|
Manufacturer: (Name and address)
|
Minimum a.1.% purity
|
a.i. Range %
|
|
5. FORMULATION
|
|
5.1 Formulator: (Name)
|
Postal AddressPhysical address
|
|
Internal code
|
|
(8) Composition (Information on composition may be attached in sealed envelope)
|
|
|
Ingredients and Function
|
Units (w/w, w/v etc.)
|
Units (e.g. cfu or IUP
|
Range
|
|
|
|
6. BIOLOGICAL PROPERTIES OF ACTIVE AGENT
|
|
6.1 History and geographical distribution of active agent
|
|
|
6.2 Mode of action and host range
|
|
|
6.3 Life cycle
|
|
|
6.4 Infectivity, dispersal and colonizing ability
|
|
|
6.5 Relationships to know plant, animal or human pathogens
|
|
|
6.6 Genetic stability
|
|
|
6.7 Information on the production of metabolites, especially antibiotics and toxins
|
|
|
7. TOXICOLOGY (active agent)
|
|
7.1 Rat
|
Acute Oral(LD 50 mg/kg)
|
Inhalation LC50 (mg/4/hour
|
Intra-peritoneal injection for infectivity (LD 50 g/kg)
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
|
Calculated
|
Calculated
|
Calculated
|
|
Hypersensitivity/allergiesin humans
|
|
|
|
|
8. TOXICOLOGY (formulated product)
|
|
|
Acute Oral (LD50 mg/kg)
|
Acute Dermal(LD50 g/kg)
|
Inhalation LC50 (mg/4/hour)
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
|
Calculated
|
Calculated
|
Calculated
|
|
8.2 Rabbit
|
Skin Irritation
|
Eye Irritation
|
|
None
|
|
|
|
Mild
|
|
|
|
Moderate
|
|
|
|
Severe
|
|
|
|
8.3 Skin Sensitization in guinea pig: (tick)
|
None
|
Mild
|
Moderate
|
Severe
|
|
8.4 WHO classification (tick)
|
la
|
lb
|
II
|
III
|
Others
|
|
8.5 Summary of other mammalian toxicological studies: e.g. livestock, wildlife, poultry, pets
|
|
|
9.ECOTOXICOLOGY
|
|
9.1 Toxicity to bees:
|
|
|
9.2 Toxicity to fish and other aquatic organisms
|
|
|
9.3 Toxicity to birds
|
|
|
9.4 Toxicity to earthworms or other soil invertebrates and soil micro-organisms
|
|
|
9.5 Toxicity to other non-target organisms
|
|
|
9.6 Persistence in environment
|
|
|
9.7 Available toxicological data relating to other ingredients in formulation (non-active additives in formulation)
|
|
|
9.8 Other effects: Specify
|
|
|
10. PACKAGING
|
|
10.1 Packaging material/container
|
|
|
10.2 Pack size(s)
|
|
|
10.3 Disposal of empty container (s)
|
|
|
11. OTHER SPECIFIC REQUIREMENTS
|
|
11.1 Operator exposure
|
|
|
11.2 Sanitary and phytosanitary measures
|
|
|
11.3 Has the product been cleared by the phytosanitary authorities? (tick)a. In the country of originb. the recipient country
|
Yes ☐(provide evidence)No ☐(give reasons)
|
|
12. DECLARATION
|
|
For and on behalf of ...................................................................I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete.
|
|
..............................................Name in full (printed).................................................Official Title
|
...............................................................Signature................................................................Date
|
_____________________________
|
FORM A2
|
(r. 4C(1))
|
|
APPLICATION FOR REGISTRATION OF A MICROBIAL BIOPESTICIDE (PEST CONTROL PRODUCTS-MACROBIOLOGICAL AGENT)
|
|
PRODUCT TRADE NAME ..............................................
|
|
PURPOSE OF APPLICATION (tick as appropriate) ......................
|
|
(a) Biopesticides containing a new active agent
|
|
|
(b) Biopesticides where source of active and/or formulation is not identical to that of a registered product
|
|
|
(c) Registration transfer
|
|
|
(d) Amendments to existing registration
|
|
|
(e) Other (Explain)
|
|
|
Will the product be marketed under own label Yes ☐ No ☐If No, specify.......................
|
|
Proposed date of marketing....................................................................................................................................
|
|
1. APPLICANT
|
|
|
1.1 Name of applicant
|
|
|
1.2 Corporate name of company
|
|
|
1.3 Reg. No. of the company
|
|
|
1.4 Name of registration holder
|
|
|
1.5 Name of local agent in country (If different from registration holder)
|
|
|
1.6 Status : (Importer/formulator/distributor etc.)
|
|
|
1.7 Physical Address
|
1
|
2
|
|
1.8 Postal Address
|
1
|
2
|
|
1.9 Telephone (and area code):
|
1
|
2
|
|
Fax (and area code):
|
1
|
2
|
|
E-mail
|
1
|
2
|
|
2. PRODUCT
|
|
2.1 Identity and stage(s) of active agent and culture collection code
|
|
|
2.2 Concentration of active agent in technical material.
|
|
|
2.3 Description of product.
|
Trade name
|
|
Trade mark
|
|
Trade mark holder
|
|
Internal code
|
|
2.4 Function of the product: (e.g. predator, parasitoid, entomopathogenic nematode).
|
|
|
2.5 Intended use: (veterinary, horticultural, public health, industrial, agriculture, forestry, etc.).
|
|
|
2.6 Target pest(s) and host (s).
|
|
|
2.7 Method, dosage rates and frequency of application.
|
|
|
2.8 Type of formulation: (if any).
|
|
|
2.9 Is the product registered in country of:
|
|
(a) origin
|
Yes ☐ If no, specify ..................................
|
No ☐
|
|
(b) manufacture
|
Yes ☐If no, specify ..................................
|
No ☐
|
|
(c) formulation
|
Yes ☐If no, specify ..................................
|
No ☐
|
|
2.10 Registration in SEARCH country(ies):(country names, product name and registration number).
|
|
|
2.11 Registration in other country (ies), particularly OECD countries: (country names, product name and registration number)
|
|
|
2.12 Customs Tariff Code: (Brussels Tariff Nomenclature).
|
|
|
3. IDENTIFICATION
|
|
Identification of Microbiological agent
|
Life stage (egg/adult/larva etc)
|
|
3.1 IdentificationScientific nameCommon name(s)
|
Genus
|
Species
|
Sub species
|
|
|
|
|
|
|
|
3.2 Contents (number per Unit)
|
|
|
4. SOURCE
|
|
Source (original isolation)
|
|
|
5. FORMULATION
|
|
|
5.1 Formulator: (Name)
|
Postal Address:Physical address:
|
|
5.2 Internal code
|
|
5.3 Composition (information on composition may be attached in sealed envelop)
|
|
Ingredients and Function
|
Units
|
Range
|
|
|
|
|
6. SUMMARY OF ENVIRONMENTAL EFFECTS (BIOSAFETY)
|
|
6.1 Risk assessment for replacement of indigenous or endangered species in same niche (exotic microbials only)
|
|
6.2 Risk to bees
|
|
|
6.3 Risk to fish and other aquatic organisms
|
|
|
6.4 Risk to birds
|
|
|
6.5 Risk to earthworms and soil micro-organisms
|
|
|
6.6 Risk to other non-target organisms
|
|
|
6.7 Other effects : specify (human health problems)
|
|
|
7. PACKAGING
|
|
|
7.1 Packaging material/container
|
|
|
7.2 Pack size(s)
|
|
|
8. OTHER SPECIFIC REQUIREMENTS
|
|
|
8.1 Operator exposure
|
|
|
8.2 Likely operator exposure under field conditions
|
|
|
8.3 Sanitary and phytosanitary measures
|
|
|
8.4 Has the product been cleared by the phytosanitary authorities?
|
Yes ☐ No ☐
|
|
9. DECLARATION
|
|
For and on behalf of ....................................................... I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete
|
|
..............................................Name in full (printed
|
...................................................................Signature
|
|
.............................................Official Title
|
....................................................................Date
|
NOTE: The format of this application form is recognized by all SEARCH countries.
_____________________________
|
FORM A3
|
(r. 4D)
|
|
APPLICATION FOR REGISTRATION OF A BIOCHEMICAL PESTICIDE (PEST CONTROL PRODUCT-BIOCHEMICAL PRODUCTS)
|
|
PRODUCT TRADE NAME .........................................................................................
|
|
PURPOSE OF APPLICATION (tick as appropriate) .................................................................
|
|
a. Biochemical pesticides containing a new active ingredient ☐
|
|
b. Biochemical pesticides where source of active and/or formulation is not identical to that of a registered product ☐
|
|
c. Registration transfer ☐
|
|
d. Amendments to existing registration ☐
|
|
e. Other (Explain)............................................................................................................................................................................................................................................................................................................
|
|
Will the product be marketed under own label Yes ☐ No ☐
|
|
If no, specify .........................................................................................................................
|
|
1. APPLICANT
|
|
1.1 Identification
|
|
|
1.2 Name of applicant/Corporate name of company
|
|
|
1.3 Reg No.
|
|
|
1.4 Name of registration holder
|
|
|
1.5 Name of local agent in country (if different from registration holder)
|
|
|
1.6 Status (Importer/formulator/distributor etc.)
|
|
|
|
1.7 Physical Address
|
|
|
|
1.8 Postal Address
|
|
|
|
1.9 Telephone (and area code)
|
|
|
|
1.10 Fax (and area code)
|
|
|
|
1.11 E-mail
|
|
|
|
2. PEST CONTROL PRODUCTS
|
|
|
2.1 Identify
|
|
|
2.2 Concentration of a.i.
|
|
|
2.3 Designation(Description of product)
|
Trade name
|
|
Trade mark
|
|
Trade mark holder
|
|
Internal code
|
|
2.4 Function of product (e.g. Insecticide, herbicide etc.)
|
|
|
2.5 Intended use (Veterinary, public health, industrial, agricultural, forestry etc.)
|
|
|
2.6 Target pest (s) and host (s)
|
|
|
2.7 Method, dosage rates and frequency of application
|
|
|
2.8 Types of formulation (e.g. EC, WP, etc.)
|
|
|
|
|
2.9 Is the product registered in country of
|
|
|
(a) origin
|
Yes ☐
|
No ☐
|
|
|
If no, specify....................................
|
|
(b) manufacture
|
Yes ☐
|
No ☐
|
|
|
If no, specify..........................
|
|
(c) formulation
|
Yes ☐
|
No ☐
|
|
|
If no, specify...................
|
|
2.10 Registration in SEARCH** country/ies (names)
|
|
|
2.11 Registration in other country/ies, especially OECD countries (names)
|
|
|
2.12 Custom Tariff Code (Brussels Tariff Nomenclature)
|
|
*Formerly GCPF.
**SEARCH-Southern and Eastern African Regulatory Committee on Harmonization of Pesticide Registration
|
3. COMPOSITION OF ACTIVE INGREDIENT (S) (Technical grade) (Information on a.i may be attached in sealed envelope)
|
|
Active ingredient (s):(Common name/s)
|
Manufacturer: (Name and address)
|
Minimum a.i.%
|
Purity a.i. Range %
|
|
4. TOXOCOLOGY OF ACTIVE INGREDIENTS(Technical grade)
|
Acute Oral(LD50 mg/kg)
|
Acute dermal (LD50 mg/kg)
|
InhalationLC50 (mg/1/hour
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
|
Calculated
|
Calculated
|
Calculated
|
|
|
|
|
|
|
5. FORMULATION
|
|
|
|
|
5.1 Formulator: (Name)
|
Postal Address
|
|
5.2 Internal code:
|
Physical Address
|
|
5.3 Composition (Information on compensation may be attached in sealed envelope)
|
|
Ingredients and Function
|
Units
|
Units
|
Range
|
|
6. TOXICOLOGY (formulated product)
|
|
6.1 Rat
|
Acute Oral(LD50 mg/kg)
|
Acute Demal(LD50 g/kg)
|
Inhalation LC50(mg/1/kg)
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
|
Calculated
|
Calculated
|
Calculated
|
|
6.2 Rabbit
|
Skin irritation
|
Eye irritation
|
|
|
None
|
|
|
|
|
Mild
|
|
|
|
|
Moderate
|
|
|
|
|
Severe
|
|
|
|
|
6.3 Skin Sensitization in guinea pig(tick)
|
None ☐
|
Mild ☐
|
Moderate ☐
|
Severe ☐
|
|
|
la
|
lb
|
II
|
III
|
Others
|
|
6.5 Summary of other mammalian toxicological information may be required
|
|
6.6 Summary of environmental effects
|
|
6.6.1 Toxicity to bees
|
|
|
6.6.2 Toxicity to fish and other aquatic organisms
|
|
|
6.6.3 Toxicity to birds
|
|
|
6.6.4 Toxicity to earthworms and soil micro-organisms
|
|
|
6.6.5 Toxicity to other non-target organisms may be required
|
|
|
6.6.6 Persistence in environment
|
|
|
6.6.7 Other effects: Specify
|
|
|
7. PACKAGING
|
|
7.1 Packaging material/container
|
|
|
7.2 Pack size (s)
|
|
|
7.3 Disposal of empty container(s)
|
|
|
8. OTHER SPECIFIC REQUIREMENTS
|
|
8.1 Operator exposure
|
|
8.2 Dermal absorption
|
|
8.3 Likely operator exposure under field conditions
|
|
8.4 Available toxicological data relating to other ingredients in formulation (non-active additives in formulation)
|
|
9. DECLARATION
|
|
|
For and on behalf of ................................................ I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete.
|
|
.................................................................................Name in full (printed)
|
.....................................................Signature
|
|
...................................................................................Official Title
|
......................................Date
|
|
Official Stamp ofApplicant/ Company
|
FOR OFFICIAL USERemarks ...........................................................................................................................................................................Signed ........................... Date
|
NOTE: The format of this application is recognized by all SEARCH countries.
_____________________________
|
FORM A3a
|
(r. 4D(4), (5))
|
|
APPLICATION FOR THE REGISTRATION OF A SEMIOCHEMICAL PEST CONTROL PRODUCT
|
|
|
Introduction
| 1. |
These guidelines are for any proposed use of the semichemicals (e.g. Pheromone, Allomone, Kairomone, Sysndrome, etc) of naturally occuring organisms for the monitoring and control of pests, livestock and public health.
|
| 2. |
Information in support of a request for registration, both published and unpublished should be supplied in the form of a summary data sheet laid out according to the format given in Form A3a.
|
| 3. |
List II of this form applies to a semiochemical not physically combined with a pesticide. Where a semiochemical is physically combined with a synthetic insecticide then the requirements under list II of Form A will apply.
Where a semiochemical and an insecticide are used separetly in a device then the requirements under List I and II in Form A will apply for the insecticide.
|
| 4. |
A pre-registration consultation between the applicant and the registration authority is strongly recommended.
Information for Applicants
|
| 1. |
The application form must be completed by a person duly authorized by the applicant/company
|
| 2. |
The application form must be submitted in triplicate to:
The Secretary, Pest Control Products Board (PCPB) P.O. BOX. 13794, 00800 Nairobi.
Email address:pcpboard@todays.co.ke/md@pcpb.or.ke Tel: 254-020- 4446115/4450242
Fax: 254-020-4449072
|
| 3. |
Every application must be accompanied by-
| (a) |
registered fee as prescribed.
|
| (b) |
three copies of the draft label as per PCPB requirements.
|
|
| 4. |
The applicant shall be required to submit:-
| (a) |
a sample of the pest control product;
|
| (b) |
a sample of the techical grade of its active ingredient.
|
| (c) |
a sample of the reference standard of its active ingredient of known purity.
|
| (d) |
any other sample as may be required by PCPB.
|
|
| 5. |
List I and II are supplied as check list and an index to ensure that the applicant has provided all relevant data.
|
| 6. |
The application must be accompanied by a tecnical dossier as per PCPB data requirements i.e. Lists I and II.
|
| 7. |
An applicant who is not a resident in Kenya must appoint an agent permanently resident in Kenya.
|
PURPOSE OF APPLICATION (tick as appropriate)
|
a. Semiochemical pest control product containing a new active ingredient ☐
|
|
b. Semiochemical pest control product where source of active and/or formulation is not identical to that of a registered product ☐
|
|
c. Registration transfer ☐
|
|
d. Amendments to existing registration ☐
|
|
e. Other (Explain) ........................................................................................................................................................................................................................................................
|
|
Will the product be marketed under own label Yes ☐ No ☐If no, specify...........................................
|
|
|
|
1. APPLICANT
|
|
1.1 Identification
|
|
|
Name of applicant/ Corporate name of the company
|
|
|
Registration Number.
|
|
|
Name of registration holder.
|
|
|
Name of local agent i country: (if different from registration holder)
|
|
|
1.2 Status:(Importer/formulator/distributor) etc.
|
|
|
|
1.3 Physical Address
|
|
|
|
1.4 Postal Address
|
|
|
|
1.5 Telephone: (and area code)
|
|
|
|
1.6 Fax:(and area code)
|
|
|
|
2. PRODUCT
|
|
2.1 Identity
|
|
|
2.2 Concentration of a.i.
|
|
|
2.3 Designation(Description of product)
|
Trade name:
|
|
|
Trade mark:
|
|
|
Trade mark holder:
|
|
|
Internal code:
|
|
2.4 Function of product:(e.g. attractant, repellent, Mating disruptor, etc.)
|
|
|
2.5 Intended use: (Veterinary, public health, industrial, agriculture, forestry, etc.
|
|
|
2.6 Target pest(s) and host(s)
|
|
|
2.7 Method, dosage, rates and frequency of application:
|
|
|
2.8 Types of formulation: (e.g. EC,WP, etc.)
|
|
|
|
|
FOR INFORMATION
|
|
|
2.9 Is the product registered in country of(a) origin:(b) manufacture:(c) formulation:
|
Yes ☐ No ☐If no, specify..............................Yes ☐ No ☐If no, specify............................Yes ☐ No ☐If no, specify..........................
|
|
2.10 Registration in SEARCH* countries/ies: (names)
|
|
|
2.11 Registration in other country/ies, especially OECD**countries: (names)
|
|
|
2.12 CustomsTariff Code:(Brussels Tariff Nomeclature)
|
|
*SEARCH- Southern and Eastern African Regulatory Committee on Harmonisation of Pesticide Registration
** OECD- Organisation for Economic Cooperation Development
|
3. COMPOSITION OF ACTIVE INGREDIENT(S) (Technical grade) (Information on a.i may be attached in a sealed envelope)
|
|
Active ingredient (s):(Common name(s)
|
Manufacturer:(Name and address)
|
Minimum a.i% purity
|
a.i% Range%
|
|
4. TOXICOLOGY OF ACTIVE INGREDIENTS (Technical grade)(Exempt for SCLPS2)
|
Acute Oral(LD50 mg/kg)
|
Acute dermal (LD50 mg/kg)
|
Inhalation LC50 mg/1/hour)
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
5. FORMULATION
|
Calculated
|
Calculated
|
Calculated
|
|
5.1 Formulator: (Name)
|
Postal Address:Physical address:
|
|
5.2 Internal code:
|
|
5.3 Composition (Information on composition may be attached in sealed envelope
|
|
Ingredients and Function:
|
Units
|
Units
|
Range
|
|
6. TOXICOLOGY (formulated product)(Exempt for SCLPs if inert are of known toxicity)
|
|
6.1 Rat:
|
Acute Oral (LD50 mg/kg)
|
Acute Dermal LD50 g/kg)
|
Inhalation LC50 (mg/1/hour)
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
|
Calculated
|
Calculated
|
Calculated
|
|
6.2 Rabbit:
|
Skin irritation
|
Eye irritation
|
|
None
|
|
|
|
Mild
|
|
|
|
Moderate
|
|
|
|
Severe
|
|
|
|
6.3Skin Sensitisation in guinea pig (tick)
|
None Mild Moderate Severe☐ ☐ ☐ ☐
|
|
6.4 WHOclassification:
|
Ia
|
Ib
|
II
|
III
|
Others
|
|
6.5 Summary of other mammalian toxicological information may be required
|
|
6.6 Summary of environmental effects
|
|
6.6.1 Toxicity to bees:
|
|
|
6.6.2 Toxicity to fish:
|
|
|
6.6.3 Toxicity to birds:
|
|
|
6.6.4 Toxicity to earthworms:
|
|
|
6.6.5 Toxicity to other non-target organisms may be required:
|
|
|
6.6.6 Other effects: Specify
|
|
|
PACKAGING
|
|
Type of packaging (packaging material/container, compatability with content)
|
|
|
7.1 Pack size (s)
|
|
|
7.2 Manner of packaging
|
|
|
7.3 Specification for primary packaging
|
|
|
7.4 Disposal of empty container (s):
|
|
Please note that the product must be sold only in the package size and type notified to the Pest Control Products Board and for which the label is approved.
| 8. |
OTHER SPECIFIC REQUIREMENTS
|
8.1 Operator exposure
|
|
8.2 Likely operator exposure under field conditions
|
|
8.3 Available toxicological data relating to other ingredients in formulation (non-active additives in formulation).
|
|
9. DECLARATION
|
|
|
For and on behalf of................................................. I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete.
|
|
...............................................Name in full (printed)
|
..............................Signature
|
|
........................................Official Title
|
...................................Date
|
|
Official Stampof Applicant/Company
|
FOR OFFICIAL USERemarks.............................................................................................................................................. ...................Signed: Date
|
NOTE: The format of this application form is recognized by all SEARCH countries.
|
_____________________________
|
FORM A3b
|
(r. 4(1), 4D(5))
|
|
APPLICATION FOR INTRODUCTION OF A STRAIGHT CHAIN LEPIDOPTERAN PHEROMONE FOR PEST MONITORING PURPOSES
|
|
|
Introduction
| 1. |
These guidelines are for any proposed use of the straight chain lepidopteran pheromones (SCLP) of naturally occurring organisms for the monitoring and control of pests of crops, livestock and public health.
|
| 2. |
Information in support of a request for introduction, both published and unpublished should be supplied in the form of a summary data sheet laid out according to the format given in Form A3b.
|
| 3. |
This form applies to a SCLP not physically combined with a pesticide. Where a SCLP is physically combined with an insecticide then the requirements under List II of Form A will apply.
|
| 4. |
Where a SCLP and an insecticide are used separately in a device then the requirments under Lists I and II in Form A will apply for the insecticide.
|
| 5. |
A pre-introduction consultation between the applicant and the regulatory authority is strongly recommended.
Information for Applicants
|
| 1. |
The application form must be completed by a person duly authorized by the applicant/company
|
| 2. |
The application form must be submitted in triplicate to:
The Secretary, Pest Control Products Board (PCPB) P.O Box 13794, 00800 Nairobi.
E-mail address: pcpboard@todays.co.ke/md@pcpb.or.ke Tel: 254-020-4446115/4450242
Fax: 254-020-4449072
|
| 3. |
Every application must be accompanied by:-
| (a) |
introduction fee as prescribed.
|
| (b) |
three copies of the draft label as per PCPB requirements.
|
|
| 4. |
The applicant shall be required to submit-
| (a) |
a sample of the pest control product;
|
| (b) |
a sample of the reference standard of its ingredient of known purity.
|
| (c) |
any other sample as may be required by PCPB.
|
|
| 5. |
An applicant who is not a resident in Kenya must appoint an agent permanently resident in Kenya.
|
PURPOSE OF APPLICATION (tick as appropriate)
|
a. SCLP pest control product containing a new active ingredient ☐
|
|
b. SCLP pest control product where source of active and/or formulation is identical to that of an existing product ☐
|
|
c. Amendments to existing product ☐
|
|
d. Other (Explain) ...........................................................................................................................................................................................................................................................................
|
|
1. APPLICANT
|
|
1.2 Identity
|
|
|
Name of applicant/Corporate name of company
|
|
|
Registration Number of company:
|
|
|
Name of registration holder of product.
|
|
|
Name of local agent in country:(if different from registration holder)
|
|
|
1.3 Status:(Importer/formulator/distributor)etc.
|
|
|
|
1.4 Physical Address
|
|
|
|
1.5 Postal Address:
|
|
|
|
1.6 Telephone:(and area code)
|
|
|
|
1.7 Fax (and area code):
|
|
|
|
2. PRODUCT
|
|
2.15 Identity (name(s) of a.i
|
|
|
2.16 Concentration of a.i.
|
|
|
2.17 Designation(Description of product)
|
Trade name:
|
|
|
Trade mark:
|
|
|
Trade mark holder:
|
|
|
Internal code:
|
|
2.18 Function of product:
|
Is it an attractant? Yes ☐ No ☐
|
|
2.19 Intended use: (Veterinary, public health, industrial agriculture, forestry, etc.)
|
|
|
2.20 Target pest (s) and host (s)
|
|
|
2.21 Method of use, frequency of replacements of the SCLP and No. of devices per unit area.
|
|
|
2.22 Degree of species specificity
|
|
|
2.23 Time of application
|
|
FOR INFORMATION
|
2.24 Is the product used in country of(a) origin:(b) manufacture:(c) formulation:
|
Yes ☐ No ☐If yes, attach evidence.............................If no, explain............................................Yes ☐ No ☐If yes, attach evidence...............................If no, explain.......................................Yes ☐ No ☐If yes, attach evidence ...........................If no, explain ....................................
|
|
2.25 Use in SEARCH*country/ies:(names)
|
If yes, attach evidence ......................
|
|
2.26 Use in other country/ies, especially OECD** COUNTRIES: (names)
|
If yes, attach evidence ...................................
|
|
2.27 Customs Tariff Code (Brussels Tariff Nomenclature):
|
|
*SEARCH — Southern and Eastern African Regulatory Committee on Harmonisation of Pesticide Introduction
**OECD- Organisation for Economic Cooperation and Development
|
3. COMPOSITION OF ACTIVE INGREDIENT(S) (Technical grade) (Information on a.i may be attached in a sealed envelope)
|
|
Active ingredient(s):(Common name/s)
|
Manufacture:(Name and address)
|
Minimum a.i.%purity
|
a.i. Range%
|
|
|
|
|
3.1 Molecular structures (to be attached)
|
|
|
3.2 Molecular formula
|
|
|
3.3 Mode of action
|
|
|
5. FORMULATION
|
|
5.1 Formulator: (Name)
|
Postal Address:
|
|
5.2 Internal code:
|
Physical address:
|
|
5.3 Composition (Information on composition may be attached in sealed envelope)
|
|
Ingredients and Function:
|
units
|
units
|
Range
|
|
5.4 Information on storage stability
|
|
|
5.5 Information on shelf life
|
|
|
5.6 Information on use and efficacy
|
|
|
|
|
|
6. Method and certificate of Analysis (to be attached)
|
|
PACKAGING
|
|
Type of packaging (packaging material/container,
compatability with content)
|
|
|
Pack size (s)
|
|
|
Manner of packaging
|
|
|
Specification for primary packaging
|
|
|
7.4 Disposal of empty container (s):
|
|
Please note that the product must be sold only in the package size and type notified to the Pest Control Products Board.
|
8. INFORMATION ON DEVICE (e.g. TRAP)
|
|
8.1 Type
|
|
|
8.2 Weight
|
|
|
8.3 Thickness/height
|
|
|
8.4 Diameter/size
|
|
|
8.5 Colour
|
|
| 9. |
OTHER SPECIFIC REQUIREMENTS
|
9.1 Operator exposure
|
|
9.2 Available toxicological data relating to other ingredients in formulation (non-active additives in formulation).
|
|
10. DECLARATION
|
|
|
For and on behalf of........................................... I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete.
|
|
........................................................Name in full (printed)
|
................................................Signature
|
|
............................................................Official Title
|
................................................Date
|
|
Official Stampof Applicant/ Company
|
FOR OFFICIAL USERemarks...............................................................................................................................................................................................................................Signed: .......... Date: ..........
|
NOTE: The format of this application form is recognized by all SEARCH countries.
|
_____________________________
|
FORM A4
|
(r. 4A(2) )
|
|
APPLICATION FOR THE REGISTRATION OF A PEST CONTROL PRODUCT (GENERIC)
|
|
|
Introduction
These guidelines are for registration of identical products that are manufactured after the expiry of the patent of an original/proprietary registered product.These identical products are generally referred to as generics and will include conventional and biochemical pesticides. A pre-registration consultation between the applicant and the registration authority is strongly recommended.
Information for Applicants
| 1. |
The application form must be completed by a duly authorized person.
|
| 2. |
The application must be submitted in triplicate to:
The Managing Director/Secretary
Pest Control Products Board (PCPB)
P.O. Box 13794-00800 Nairobi.
E-mail address: pcpboard@todays.co.ke/md@pcpb.or.ke
Tel: 254-020-8021846/7/8 Fax: 254-020-8021865
Website: www.pcpb.or.ke
|
| 3. |
Every application must be accompanied by:-
a) application fee as prescribed (Registration fee is payable upon approval by the Board).
b) 3 copies of the draft label as per PCPB requirements.
|
| 4. |
The applicant may be required to submit to:-
| (a) |
a sample of the pest control product;
|
| (b) |
a sample of the technical grade of its active ingredient;
|
| (c) |
a sample of the laboratory standard of its active ingredient;
|
| (d) |
any other sample as may be required by the Board.
|
|
| 5. |
List I and II are supplied as a check list and an index to ensure that the applicant has provided the relevant data.
|
| 6. |
The application must be accompanied by a technical dossier as per PCPB data requirements (Lists I and II)
|
| 7. |
An applicant who is not a resident in Kenya must appoint an agent permanently resident in Kenya and duly recognized by the Pest Control Products Board.
TRADE NAME ...........................
PURPOSE OF APPLICATION (tick/fill as appropriate)
|
a. Pest control product containing a generic active ingredient ☐i) Date of expiry of patent.............................................ii) Name of former patent holder....................................
|
|
b. Pest control product where source of active and/or formulation is not identical to that of a registered product ☐
|
|
c. Registration transfer ☐
|
|
d. Amendments to existing registration (e.g inerts, source of technical material e.t.c) ☐
|
|
e. Other (Explain)....................................................................................................................................................................................................................
|
|
f. Will the product be marketed under own label? Yes ☐ No ☐If no specify........................................................................
|
|
Proposed date of marketing..............................................
|
|
1. APPLICANT
|
|
1.1 Identification
|
|
|
Name of applicant/Corporate name of company
|
|
|
Business Registration No:
|
|
|
Name of registration holder
|
|
|
1.12 Status: (Manufacturer formulator/other)
|
|
|
1.13 Physical Address
|
|
|
1.14 Postal Address:
|
|
|
1.15 Telephone: (and area code)
|
|
|
1.16 Fax:(and area code)
|
|
|
1.17 e-Mail:
|
|
|
2. Name of local agent in country:(if different from registration holder)
|
|
|
Business Registration No:
|
|
|
2.1 Status:(Importer/formulator/distributor)
|
|
|
2.2 Physical Address
|
|
|
2.3 Postal Address
|
|
|
2.4 Telephone:(and area code)
|
|
|
2.5 Fax:(and area code)
|
|
|
2.6 e-mail:
|
|
|
3 PRODUCT
|
|
Designation(Description of product)
|
Trade name:
|
|
|
Trade mark:
|
|
|
Trade mark holder:
|
|
Function of product: (eg. Insecticide, herbicide etc.)
|
|
|
3.1 Intended use: (Veterinary, public health, industrial
|
|
|
3.2 Target pest(s) and host (s)
|
|
|
3.3 Method, dosage rates and frequency of application:
|
|
|
3.4 Type of formulation: (eg. EC, WP, etc.)
|
|
Crop Life International(CLI) Code (if available)
|
|
|
3.5 a) Is the technical grade (active ingredient) registered in country of manufacture?
|
Yes ☐ No ☐If no, give reasons ..........................
|
|
b) Is the product registered in the country of formulation
|
Yes ☐ No ☐If no, give reasons ..........................
|
|
3.6 Proof of registration in SEARCH** country/ies (names)
|
|
|
3.7 Proof of registration in other countries.
|
|
|
3.8 Customs Tariff Code:(Brussels Tariff Nomenclature)
|
|
|
4 COMPOSITION OF ACTIVE INGREDIENT (S) (Technical grade) (Information on a.i may be attached in sealed envelope
|
|
Active ingredient (s):(Common name/s)
|
Manufacturer:(Name and address)
|
Minimum a.i.%purity
|
a.i. Range%
|
|
FORMULATION
|
|
5.1 Formulator: (Name)Postal Address:Physical address:
|
|
5.2 Internal code:
|
|
5.3 Composition (Information on composition may be attached in sealed envelope)
|
|
Ingredients and Function:
|
g/L
|
g/Kg
|
Range
|
|
|
|
|
|
|
6. TOXICOLOGY (formulated product)
|
|
6.1 Rat:
|
Acute Oral (LD50 mg/Kg)
|
Acute Dermal(LD50 mg/Kg
|
Inhalation LC50 (mg/L/4 hour
|
|
|
Experimental
|
Experimental
|
Experimental
|
|
6.2 Rabbit:
|
Skin Irritation
|
Eye irritation
|
|
None
|
|
|
|
Mild
|
|
|
|
Moderate
|
|
|
|
Severe
|
|
|
|
6.3 Skin Sensitization in guinea pig: (tick)
|
None ☐ Mild ☐ Moderate ☐ Severe ☐
|
|
6.4 WHO
|
Ia
|
Ib
|
II
|
III
|
Table V
|
|
7 Summary of environmental effects
|
|
7.1 Toxicity to bees:
|
|
|
7.2 Toxicity to fish and other aquatic organisms:
|
|
|
7.3 Toxicity to birds:
|
|
|
7.4 Toxicity to earthworms and soil micro-organisms:
|
|
|
7.5 Toxicity to other non-target organisms:
|
|
|
7.6 Persistence in environment:
|
|
|
7.7 Other effects: Specify
|
|
|
8. PACKAGING
|
|
8.1 Packaging material/container:
|
|
|
8.2 Pack size (s):
|
|
|
8.3 Disposal of empty container (s):
|
|
|
9. OTHER SPECIFIC REQUIREMENTS
|
|
9.1 Operator exposure
|
|
9.1.1 Dermal absorption.
|
|
9.1.2 Likely operature exposure under field conditions
|
|
9.2 Available toxicological data relating to other ingredients in formulation (non-active additives in formulation)
|
|
10. DECLARATION
|
|
For and on behalf of ............................................ I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete.
|
|
.....................................................Name in full (printed)....................................................Signature
|
|
.............................................Official Title............................................DateOfficial Stampof Applicant/ Company
|
|
FOR OFFICIAL USERemarks.................................................................................................................................................................................................................................
|
|
.............................................Signed............................................Date
|
NOTE: The format of this application is recognized by all SEARCH countries.
|
_____________________________
APPLICATION FOR THE REGISTRATION OF A PEST CONTROL PRODUCT (SPRAY ADJUVANT)
A spray adjuvant: Is a compound or substance than enhances or modifies or is intended to enhance or modify the physical or chemical characteristics of a pest control product to which it is added.
Information for Applicants
| 1. |
The application form must be completed by a duly authorized person.
|
| 2. |
The application must be submitted in triplicate to:
The Managing Director/Secretary
Pest Control Products Board (PCPB)
P.O. Box 13794-00800 Nairobi.
E-mail address: pcpboard@todays.co.ke/md@pcpb.or.ke
Tel: 254-020-8021846/7/8 Fax: 254-020-8021865
Website: www.pcpb.or.ke
|
| 3. |
Every application must be accompanied by:-
| (a) |
application fee as prescribed (Registration fee is payable upon approval by the Board).
|
| (b) |
3 copies of the draft label as per PCPB requirements.
|
|
| 4. |
The applicant may be required to submit;-
| (a) |
a sample of the pest control product;
|
| (b) |
a sample of the laboratory standard of its active ingredients;
|
| (c) |
any other sample as may be required by the Board.
|
|
| 5. |
List I is supplied as a check list and an index to ensure that the applicant has provided the relevant data.
|
| 6. |
The application must be accompanied by a technical dossier as per PCPB data requirements(dossier index.)
|
| 7. |
An applicant who is not a resident in Kenya must appoint an agent permanently resident in Kenya and duly recognized by the Pest Control Products Board.
TRADE NAME ...............................
PURPOSE OF APPLICATION (tick as appropriate)
|
a. A pest control product which is an adjuvant ☐
|
|
b. Pest control product where source of active and/or formulation is not identical to that of a registered product ☐
|
|
c. Registration transfer ☐
|
|
d. Amendments to existing registration
|
|
e. Other (Explain) ............................................................................................................................................................................................................
|
|
f. Will the product be marked under own label? Yes ☐ No ☐If no specify ........................................................................................................
|
|
Proposed date of marketing .....................................................................
|
|
1. APPLICANT
|
|
1.1 Identification
|
|
|
Name of applicant/ Corporate name of company
|
|
|
Business Registration No:
|
|
|
Name of registration holder
|
|
|
1.18 Status: (manufacturer/ formulator/other)
|
|
|
1.19 Physical Address
|
|
|
1.20 Postal Address:
|
|
|
1.21 Telephone:(and area code)
|
|
|
1.22 Fax:(and area code)
|
|
|
1.23 E-mail:
|
|
|
2. Name of local agent in country:(if different from registration)
|
|
|
Business Registration No:
|
|
|
3.1 Status:(Importer/formulator/distributor)
|
|
|
3.2 Physical address
|
|
|
3.3 Postal Address:
|
|
|
3.4 Telephone:(and area code)
|
|
|
3.5 Fax:(and area code)
|
|
|
3.6 e-mail:
|
|
|
3. PRODUCT
|
|
|
3.1 Designation(Description of product)
|
Trade name:
|
|
|
Trade mark:
|
|
|
Trade mark holder:
|
|
3.2 Spray adjuvant function:(wetter, surfactanct, etc)
|
|
|
3.3 Intended use: (Veterinary, public health, industrial, agriculture, forestry, etc.
|
|
|
3.4 Target usee.g product and crop/animal
|
|
|
3.5 Method, dosage rates and frequency of application:
|
|
|
3.6 Type of formulation: (e.g. EC,WP, etc.)
|
|
Crop Life International (CLI) Code (if available)
|
|
|
3.7 (a) Is the technical grade (active ingredient) registered in country of manufacture?(b) Is the product registered in the country of formulation?
|
Yes ☐ No ☐If no, give reasonsYes ☐ No ☐If no, give reasons
|
|
3.8 Registration in SEARCH** country/ies: (names)
|
|
|
3.9 Proof of existing registration in other country (ies)
|
|
|
3.10 CustomsTariff Code:(Brussels Tarrif Nomenclature)
|
|
|
4. SPRAY ADJUVANT FORMULA (attach confidential formula)
|
|
Active ingredient(s):(Common name/s)
|
Manufacturer: (Name and address
|
Spray adjuvant function
|
Percentage
|
|
|
|
|
|
|
5. FORMULATION
|
|
5.1 Formulator: (Name)Postal Address:Physical address:
|
|
5.2 Internal code:
|
|
5.3 Composition (Information on composition formula may be attached in sealed
|
|
Ingredients
|
g/L
|
g/KG
|
Range
|
|
|
|
|
|
|
6. TOXICOLOGY (formulated product)
|
|
6.1 Rat:
|
Acute Oral (LD50 mg/Kg)
|
Acute Dermal (LD50 mg/Kg)
|
Inhalation LC 50 (mg/L/4/hour
|
|
|
Experimental/Calculated
|
Experimental/Calculated
|
Experimental/Calculated
|
|
6.2 Rabbit:
|
Skin irritation
|
Eye irritation
|
|
None
|
|
|
|
Mild
|
|
|
|
Moderate
|
|
|
|
Severe
|
|
|
|
6.3 Skin Sensitization in guinea pig: (tick)
|
None ☐ Mild ☐ Mode ☐ Severe ☐
|
|
6.4 WHO classification:
|
Ia
|
Ib
|
II
|
III
|
Table V
|
|
6.5 Summary of other mammalian toxicological studies: eg. Livestock, wildlife, poultry, pets
|
|
7. Summary of environmental effects (where applicable e.g. sensitive areas)
|
|
7.1 Toxicity to bees:
|
|
|
7.2 Toxicity to fish and other aquatic organisms:
|
|
|
7.3 Toxicity to birds:
|
|
|
7.4 Toxicity to earthworms and soil microorganisms:
|
|
|
7.5 Toxicity to other non-target organisms:
|
|
|
7.6 Persistence in environment:
|
|
|
7.7 Other effects: Specify
|
|
|
8 PACKAGING
|
|
8.1 Packaging material/container:
|
|
|
8.2 Pack size(s):
|
|
|
8.3 Disposal of empty container(s):
|
|
|
9. OTHER SPECIFIC REQUIRMENTS
|
|
9.1 Operator exposure
|
|
9.1.1 Dermal absorption.
|
|
9.1.2 Likely operator exposure under field conditions
|
|
9.2 Available toxicological data relating to other ingredients informulation (non-active additives in formulation).
|
|
10. DECLARATION
|
|
For and on behalf of ........................................ I hereby certify that the above mentioned information and data provided in support of this application are to the best of my knowledge true, correct and complete.
|
|
.....................................................Name in full (printed)
|
......................................Signature
|
|
.....................................................Official Title
|
....................................Date
|
|
Official Stampof Applicant/Company
|
FOR OFFICIAL USERemarks......................................................................................................................................................Signed ......................Date ......................
|
|
_____________________________
APPLICATION FOR REGISTRATION AS A LOCAL AGENT
information for Applicants
| 1. |
"Agent" means a person or company who has/that has been appointed to act on behalf of a registrant in accordance with regulation 4(2) of the Pest Control Producrs (Registration) Regulations.
|
| 2. |
The application form must be completed by a duly authorized person.
|
| 3. |
The application must be submitted to:
The Managing Director/Secretary
Pest Control Products Board (PCPB)
P.O.Box 13794-00800 Nairobi
E-mail address:md@pcpb.or.ke
Tel:254-020-8021846/7/8 Fax: 254-020-8021865
Website: www.pcpb.or.ke
|
| 4. |
Every application must be accompanied by:-
| (a) |
An original letter from the registrant,
|
| (b) |
A binding agreement entered between the registrant and the agent.
|
|
| 5. |
The applicant may be required to submit:-
| (a) |
A sample of the pest control product;
|
| (b) |
A sample of the technical grade of its active ingredient;
|
| (c) |
A sample of the laboratory standard of its active ingredient;
|
| (d) |
Any other information as may be required by the Board.
|
|
| 6. |
For each product, there can only be one local agent.
|
| 7. |
The local agent shall appoint in the distributor(s)
Product Details
Trade Name............................
Name of Manufacturer.................
Name of Registrant....................
Name of agent.........................
Name of distributor....................
Signature of applicant.................. Date ..........
Official Stamp of Applicant/Company
|
_____________________________
APPLICATION FOR CHANGE OF AGENCY.
Information for Applicants
| 1. |
"Agent" means a person or company who has/that has been appointed to act on behalf of a registrant in accordance with regulation 4(2) of the Pest Control Products (Registration) Regulations.
|
| 2. |
The application form must be completed by a duly authorized person.
|
| 3. |
The application must be submitted to:
The Managing Director/Secretary
Pest Control Products Board (PCPB)
P.O.Box 13794-00800 Nairobi.
E-mail address:md@pcpb.or.ke
Tel:254-020-8021846/7/8 Fax: 254-020-8021865
Website: www.pcpb.or.ke
|
| 4. |
Every application must be accompanied by:-
| (a) |
An original letter from the registrant,
|
| (b) |
A binding agreement entered between the registrant and the agent.
|
| (c) |
An original letter of no objection from the current agent,
|
| (d) |
Application fee of Ksh 20,000 per product (change of agency fee is payable upon approval by PCPB after meeting the other requirements),
|
| (e) |
A copy of the draft label as per PCPB requirements,
|
| (f) |
Proof of licensing of the new agent by PCPB.
|
|
| 5. |
The applicant maybe required to submit:-
| (a) |
A sample of the pest control product;
|
| (b) |
A sample of the technical grade of its active ingredient;
|
| (c) |
A sample of the laboratory standard of its active ingredient;
|
| (d) |
Any other information as may be required by the Board.
|
Trade name ..................................................
Name of Manufacturer ............................................
Name of Registrant ...............................................
Registration Number (If registered) ............ Status of registration .................
Name of former agent .......................................
Name of new agent ...........................................
Signature of applicant .................................... Date ..............
Official Stamp of Applicant/Company
For official use only
Please check whether the following documents have been provided:
Registration Department:
|
| 1. |
Has an originanl letter from the registrant been provided Yes ☐ No ☐
|
| 2. |
Has an original letter of no objection from the former agent been provided?
Yes ☐ No ☐
|
| 3. |
Has the applicant attached a copy of the draft label?
Yes ☐ No ☐
Inspection Department
|
| 4. |
Is the applicant licensed as a pesticide dealer/agent with PCPB in the current year?
☐ Yes ................... No ☐ .................... if yes indicate licence No.
Accounts:
|
| 5. |
Has the applicant paid the change of agency fee? ☐ Yes ............. ☐ No.....
Indicate Receipt Number ................... Date ..............................
|
| 6. |
Has the applicationt paid the dealers/agency license fee? ☐ Yes......☐ No......
Indicate Receipt Number................... Date...........................
|
Recommended ☐
|
Not Recommended ☐
|
Recommended ☐
|
Not Recommended ☐
|
Recommended ☐
|
Not Recommended ☐
|
|
Date ...........................................................................................Registration Officer
|
Date .......................................................................................Inspector
|
Date ......................................................................................Accountant
|
................................................................
|
Approved by Managing DirectorPest Control Products Board
|
Date ..........................................................
|
Changes effected by (IT Officer).............................. Date .....................
|
_____________________________
THE PEST CONTROL PRODUCTS (MISCELLANEOUS FORMS) SUBMISSION OF SAMPLE (S) FOR EFFICACY TESTING
This form should be filled in duplicate: Part I and II to be filled by the Applicant. The completed form MUST be accompanied with a copy of:
i. the trial permit
ii. the Material Safety Data Sheet (MSDS)
The trial sample MUST be labeled in accordance with the Pest Control Products Board guidelines for experimental labeling.
Trade name .................................................................
Formulation type .............................................................
Active ingridient (s) ........................................................
Concentration of active ingidient(s) ........................................................
Quantity of sample (Liters or grams) ........................................................................
Expiry date .......................................................................................
Number of packages ......................................................................................
REF: (Permit No. and date) .....................................................................................
Recommended storage conditions of temperature ...............................................
Name of Applicant (Local agent) ..................................................................
II) Submission details
Submitted to PCPB by:
Name ............................... Signature......................Date...........................
Delivery details
Received on behalf of PCPB by:
Name ........................... Signature .................... Date ...............
(Tick Appropriately)
| 1. |
Is the application form attached with a copy of the trial permit? YES ☐ NO ☐
|
| 2. |
Is the form attached with a copy of the Material Safety Data Sheet? YES ☐ NO ☐
|
| 3. |
Does the label conform to the PCPB guidelines for experimental labeling? YES ☐ NO ☐
Institution(s) of destination ................................................
Means of delivery ............................................................
A. PCPB personnel
Name of Person delivering ....................................................
Date of delivery .............................................................
Signature ....................................................................
B. Courier Service
Name of company .............................................................
Contact person ..............................................................
Charges (Attach receipt) ...................................................
Date of delivery ............................................................
Official stamp..............................................................
C. Receiving Institution
Name of Receiving Institution ................................................
Date of receipt .............................................................
Person receiving ............................................................
Signature .................................................................
Official Stamp .........................................................
|
_____________________________
|
FORM A9
|
(r. 7(1)(b))
|
|
APPLICATION FOR CHANGE OF TRADE NAME FOR A PEST CONTROL PRODUCT.
|
|
Information for Application
|
| 1. |
The application form must be completed by a duly authorized person/agent
|
| 2. |
The application must be submitted to:
The Managing Director/Secretary
Pest Control Products Board (PCPB)
P.O Box 13794 - 00800 Nairobi.
E-mail address: md@pcpb.or.ke
Tel: 254- 020 - 8021846/7/8 Fax: 254- 020- 8021865
Website: www.pcpb.or.ke
|
| 3. |
Every application must be accompanied by:-
| (a) |
An original consent letter from the owner of the trade name (with evidence from a trade mark registering body);
|
| (b) |
A copy of the draft label as per PCPB requirements;
|
| (c) |
Proof of licencing as a dealer with Pest Contro Products by PCPB
|
|
| 5. |
The applicant may be required to submit:
| (a) |
A sample of the technical grade of its active ingredient:
|
| (b) |
A sample of the laboratory standard of its active ingridients;
|
| (c) |
Any other information as may be required by the Board.
|
|
| 6. |
Evidence from Kenya Intellectual Property Institute (KIPI) that the new trade name is available for use.
|
| 7. |
Payment of a change of trade name fee of Kshs. 10,000
Current Trade Name.......................................................
Proposed Trade Name* .........................................................
Reason for change .............................................................
.............................................................................
..............................................................................
Stage of Registration
i) Registered (Indicate registration No.) ..............................................
(Trade name to be in use 6 months after approval to allow exhaustion of old stock)
ii) Undergoing trials (State institution carrying out trials and permit No.)
iii) Other (indicate)
Name of Manufacturer ...............................................
Name of Registrant (Proprietary owner of Technical information)
...............................................................................
Name of agent .........................................................
Signature of applicant ........................ Date ..........................
Official Stamp of Applicant/Company
For official use only
Please check whether the following documents have been provided:
Registration Department:
|
| 7. |
Has an original letter from the owner of the trade name been provided? ☐ Yes No ☐
|
| 8. |
Has the applicant attached a copy of the draft label?
☐ Yes ☐ No
|
| 9. |
Has KIPI confirmed availability of new trade name? Yes ....... No .........
|
| 10. |
Has the applicant submitted a sample of the pest control product? (not mandatory) Yes ........ No.....
Inspection Department:
|
| 11. |
Is the applicant lisensed as a pesticide dealer/agent with PCPB in the current year?
☐ Yes ................. ☐ No ..............
Indicate Receipt Number................ Date........................
|
| 13. |
Has the applicant paid the change of trade name fee? ☐ Yes... ☐ no...
Indicate Receipt Number ................ Date........................
|
| 14. |
Departmental recommendations
|
Recommended ☐
|
Not Recommended ☐
|
Recommended ☐
|
Not Recommended ☐
|
Recommended ☐
|
Not Recommended ☐
|
|
Date...............................................................................................Registration Officer
|
Date................................................................................................Inspector
|
Date...............................................................................................Accountant
|
|
* superlatives are not allowed and Trade Names should be simple, append additional information
|
|
| 15. |
Recommendation of the Technical and Registration Committess of the Board
Recommended ☐ Not Recommended ☐
Reason for rejection......................................................
Date.......................................................................
|
| 16. |
Decision by the Board of Management
Approved ☐ Not Approved ☐ Date................
Reasons for rejection.................................................
|
| 17. |
Changes affected on Lists of Products and data base by (IT Officer)
Signature....................... Date.........................
|
_____________________________
|
FORM B
|
(r. 4H)
|
|
PEST CONTROL PRODUCTS BOARD
|
The managing Director/Secretary
Pest Control Products Board (PCPB)
P.O Box 13794 - 00800 Nairobi.
E-mail address: pcpboard@todays.co.ke/md@pcpb.or.ke
Tel: 254- 020 - 8021847/7/8 Fax: 254- 020- 8021865
Website: www.pcpb.or.ke
|
SUMMARY OF THE DATA SUBMITTED TO THE PCPB FOR REGISTRATION OF A PEST CONTROL PRODUCT.
|
|
Trade Name ..................................................................................
The Name and Address of Formulator ......................................................................
Common Name of the active ingredient (s).............................................................
Concentration of active ingredients(s)....................................................................
Source of active ingredients(s)............................................................................
Chemical Name.................................................................
Formulation type..............................................................
Proposed Uses...............................................................
Packaging/Containers (Material size)......................................
Registrant (Name, Address, Status)........................................................................
Agents/Distributors in Kenya.....................................................
Premises (Reg. No. Date of issue)............................................................
| (a) |
Physical/Chemical Properties of the a.i ...................................................
|
| (b) |
Physical/Chemical properties of the technical grade material ...........................
|
| (c) |
Composition of the technical product (purity%, nature and content of impurities, isomers, by-products - other details should be provided in the dossier) ...............................................................................
|
| (d) |
Physical/Chemical Properties of the Formulated Products .........................................................................
|
| (e) |
Composition of the Formulated Product (Concentration of a.i in the formulation. other details should be provided in the dossier)................................................................................
|
| (f) |
Method of analysis for determination of the a.i in the technical and formulated products ...................................................................................
|
|
PART III
|
|
Biological(efficacy) Data
|
| (a) |
Target Pest(s), Diseases(s), Hosts(s).............................................................
|
| (b) |
Method, Rate, Frequency of application.........................
|
| (c) |
Recommendation for use in Kenya...............................................
|
| (d) |
Recommendations for use by authorized bodies outside Kenya..................................................
|
|
PART IV
|
|
Toxicological data
|
| (a) |
Acute Toxicological Data of the active ingridient(s)
...........................................................................
...........................................................................
|
| (b) |
Acute toxicity data of the formulated product:..
...........................................................................
...........................................................................
|
| (c) |
Short term toxicity studies ...............................................
|
| (d) |
Other toxicological studies:
|
| (1) |
Reproduction studies ...........................................................
|
| (2) |
Teratological studies ..........................................................
|
| (3) |
Neurotoxicity studies ..................................................................................
|
| (4) |
Mutagenecity studies .............................................................
|
| (5) |
Long term toxicity/carcinogenicity studies ......................................
|
| (6) |
Accumulation of compound in tissues ..............................................
|
| (7) |
Metabolic studies .........................................................
|
| (8) |
Effects on the livestock, poultry ..........................................
|
| (9) |
Toxicity Data on impurities .................................................
|
| (10) |
Toxicity Data on metabolites ................................................
|
| (11) |
Human toxicology and medical aspects:
|
| (1) |
Hazard to humans ...........................................................
|
| (2) |
Sysmptoms of poisoning .......................................................
|
| (3) |
Antidote ..................................................................
|
| (4) |
Treatment..................................................................
|
| (5) |
First Aid Measures .........................................................
|
| (6) |
Safety Precautions/Restrictions .............................................
TV - RESIDUE DATA
| (a) |
Principal Residues .............................................................
|
| (b) |
Disappearance and fate of residues .............................................
|
| (c) |
Method(s) of analysis (crops, soil, water, feedsuffs etc.) ....................................................................
|
PART VI
Environment and wildlife hazards
| (a) |
Degradation and mobility studies (soil, water, air) ........................................................................
|
| (b) |
Toxicity to birds ............................................................
|
| (c) |
Toxicity to fish ..........................................................
|
| (d) |
Toxicity to honeybees/beneficial insects ....................................
|
| (e) |
Toxicity to earthworms, other soil invertebrates ...............................
|
| (f) |
Changes in soil ecology ....................................................
|
Information on Approvals/Registrations in other countries
.............................................................................................................
.............................................................................................................
Draft of local label (as per Legal Notice No. 89/1984).
.............................................................................................................
.............................................................................................................
Brief prepared by .................................................................
Signature ..................................................................
Official stamp................................................................
Date ..........................................................................
PART X
Deecision of the PCPB registration Sub-Committee
Recommended/Not Recommended for registration
Reasons:-
..............................................................................................................
Date .....................................................................................
|
_____________________________
|
FORM B1
|
|
|
|
(r. 4H)
|
|
PEST CONTROL PRODUCTS BOARDP.O. BOX 13794-00800, NAIROBI, WAIYAKI WAYTel: 254-020 4446115/4450242 Fax: 254-020 4449072E-MAIL: pcpboard@todays.co.keWEBSITE ADDRESS: www.pcpb.or.ke
|
SUMMARY OF THE DATA SUBMITTEDTO THE PCPB FOR REGISTRATION OF A MICROBIAL PEST CONTROL PRODUCT.
Trade Name ............................................................
Name of the manufacturer and address ....................................
The Name and Address of Formulator ........................................
Common Name of the active Agent(s) ........................................
Concentration of the active ingridient(s) ..................................
Source of active ingridient(s) .............................................
Scientific name of the microbial Agent .....................................
Formulation type ...........................................................
Proposed Uses ..............................................................
Packaging/Containers(Material size) .........................................
Agents/Distributors in Kenya .................................................
Premises (Reg. No. Date of issue) ............................................
|
PART II
|
|
PHYSICAL/CHEMICAL PROPERTY OF THE ACTIVE AGENT
|
| (a) |
Physical/Chemical Properties of the active agent ............................................
|
.........................................................................................
| (b) |
Physical/Chemical properties of the technical grade material add (incase different from point a) above) ...................
|
.....................................................................................
| (c) |
Composition of the technical product (purity%, nature and content of impurities/contaminants, by-products - other details should be provided in the dossier)
..................................................................................
|
......................................................................................
| (d) |
Physical/Chemical Properties of the Formulated products
..................................................................................
...................................................................................
|
| (e) |
Concentration of active agents in the formulation. (Other details should be provided in the dossier) .............................................................
..................................................................................................
|
| (f) |
Method if Identification, Enumeration and Bioassay
.........................................................................................................
..........................................................................................................
|
|
PART III
|
|
BIOLOGICAL PROPERTIES OF THE MICROOGANISM
|
| (a) |
Origin of microorganisms and its uses.......................................
|
| (b) |
Effect in the non-target organisms..........................
|
| (c) |
Life cycle of the microorganisms......................
|
| (d) |
Infectivity (plant and animals)......................................
|
| (e) |
Dispersal and colonization...........................................
|
| (f) |
Effect of environmental parameters (UV, temperature, soil pH, humidity, nutrition requirements, etc.) on stability and survival
|
| (g) |
Relationships to known plant, animal or human pathogens.............................................
|
| (h) |
Genetic stability and factors affecting it (potential mutant).........................................
|
| (i) |
Information on the production of metabolites (especially toxins).......................................
|
| (j) |
Show antibiotics and other anti-microbial properties
|
|
PART IVBiological (efficacy) Data
|
| (a) |
Target Pest(s), Diseases(s), Host(s).........................................
|
| (b) |
Mode of action of the microorganism..........................................
|
| (c) |
Method, Rate, Frequency of application..........................................
|
| (d) |
Recommendations for use in Kenya..................................................
|
| (e) |
Recommendation for use by authorized bodiesoutside Kenya.............................
|
| (a) |
Acute Toxicological/Infectivity Data of the active agent(s)
.............................................................................
.............................................................................
|
| (b) |
Acute toxicity data of the formulated product:
..............................................................................
..............................................................................
|
| (c) |
Short term toxicity studies (if there is concern under Tier 1 studies) ..........................
|
| (d) |
Other toxicological studies(if concerns on Tier 1 and 2) ...........................................
|
| (1) |
Reproduction studies ..................................................................
|
| (2) |
Tertological studies .......................................................................
|
| (3) |
Neurotoxicity studies .....................................................................
|
| (4) |
Long term toxicity/carcinogenicity studies .....................................................
|
| (6) |
Metabolic studies (if microorganism organism is known to produce metabolites) .....................................................
|
| (7) |
Effects on livestock, poultry (if exposure is expected) .........................................................................
|
| (8) |
Toxicity information on impurities/contaminants if pathogenic significant .........................................................
|
| (9) |
Toxicity Data on metabolites (if applicable) ...................................
..................................................................................................
.....................................................................................................
|
| (10) |
Human toxicology and medical aspects:
|
| (1) |
Hazards to humans ......................................................................
|
| (2) |
Sysmptoms of poisoning or allergic reactions ...............................
|
| (3) |
Antidote ...........................................................................
|
| (4) |
Treatment .............................................................................
|
| (5) |
First Aid Measures ...............................................................
|
| (6) |
Safety Precautions/Restrictions ...........................................................
(Data is required if the microoganism produces metabolites)
| (a) |
Principal Residues ......................................................................
|
........................................................................................
| (b) |
Disappearance and fate of residues ....................................................
|
.....................................................................................
| (c) |
Method(s) of analysis (crops, soil, water, feedstuffs etc.).........................
.....................................................................................
....................................................................................
|
Environment and wildlife hazards
| (a) |
Degradation and mobility studies (soil, water, air) ................................
|
...........................................................................
| (b) |
Toxicity to birds .......................................................
|
| (c) |
Toxicity to fish ........................................................
|
| (d) |
Effects on aquatic-invertebrates.........................................
|
| (e) |
Toxicity to honeybees/beneficial insects.................................
|
| (g) |
Effects on other soil microorganisms .....................................
|
| (a) |
Information on Approvals by local phytosanitary authorities ....................................
|
| (b) |
registrations in other countries ...............................................................
|
| (c) |
Information on approval by national biosafety authority if GMO's
|
..........................................................................................
...........................................................................................
Draft of local label (as per Legal Notice No. 89/1984).
...............................................................................................
...............................................................................................
.
Brief prepared by..................................................................
Signature...........................................................................
Official stamp.......................................................................
Date..................................................................................
Decision of the PCPB Technical and registration Committee
Recommended/Not Recommended for registration
Reasons:-
..............................................................................................
...............................................................................................
Date ...................................................................................
|
_____________________________
|
PEST CONTROL PRODUCTS BOARDP.O BOX 13794-00800, NAIROBI, WAIYAKI WAYTel: 254-020 4449115/4450242 Fax 254-020 4449072E-MAIL: pcpboard@todays.co.keWEBSITE ADDRESS: www.pcpb.or.ke
|
|
SUMMARY OF THE DATA SUBMITTED TO THE PCPB FOR REGISTRATION OF MICROBIAL PEST CONTROL PRODUCT.
Trade Name .....................................................
Collection Number (National museum of Kenya) ...................................
The name and address of Formulator................................................
Common Name of the active agent(s)
Description of unit..........................................................
Source of active agent.......................................................
scientific name of the agent..................................................
Form of presentation (Stage of development, carrier material
.....................................................................................
Proposed Uses .....................................................................
Packaging/Containers (Material, size) ................................................................
Registrant (Name, Address, Status) ................................................................
Agents/Distributors in Kenya ...................................................................
Premises (Reg. No. Date of issue) ..................................................................
BIOLOGICAL
| (a) |
Description of the agent as presented (stage, colour, ..................)
|
| (b) |
Taxonomy ..................................................................
|
| (c) |
Descriptive identification of the agent .............................................
|
| (e) |
Host specificity range and effects on non-target species...(including invasiveness, dispersal, colonization ability) ...............................................
|
| (f) |
Development stages/life cycle ........................................................
|
| (g) |
Stability in proposed packaging ......................................................
| (i) |
Method of qualification ............................................................. |
|
| (a) |
Target Pest(s), Disease(s), Host(s)............................................
|
| (b) |
Method, Rate, Frequency of application...........................................
|
| (c) |
Recommndation for use in Kenya.......................................................
|
| (d) |
Recommendations for use by authorized bodies outside Kenya........................................................
|
| (a) |
Bio-Surveillance data available................................................
|
| (b) |
Relationships to known plant, animal or human parasites.....................................
|
| (c) |
Hazards to humans...................................................
|
| (d) |
Safety precautions/Restrictions..........................................
|
| (e) |
Recommended methods and precautions concerning handling, storage, or storage..............
|
| (f) |
Procedures for destructions.........................................
|
| (g) |
measures in cae of an accident.......................................
|
| (a) |
Effects of environmental parameters on stability and survival (IV, temperature, soil, pH, Humidity, etc)......................................
|
| (a) |
Clearance by phytosanitary authority...............................
|
| (b) |
Information on approvals/Registrations in other countries ................................
|
Draft of local label (as per Legal Notice No. 89/1984)............................................
Brief prepared by......................................................
Signature..................................................................
Official Stamp.........................................................
Date.............................................................
Decision of the PCPB registration Sub-Committee
Recommended/Not Recommended for registration
Reasons:-
............................................................................
.............................................................................
Date ........................................................
_____________________________
|
|
|
|
FORM B3
|
(r. 4H)
|
|
PEST CONTROL PRODUCTS BOARDP.O BOX 13794-00800, NAIROBI, WAIYAKI WAYTel: 254-020 4446115/1150242 Fax: 254-020 4449072E-MAIL: pcpboard@todays.co.keWEBSITE ADDRESS: www.pcpb.or.ke
|
SUMMARY OF THE DATA SUBMITTED TO THE PCPB FOR REGISTRATION OF A BIOCHEMICAL PEST CONTROL PRODUCT
| 1. |
Trade Name..............................................
|
| 2. |
The Name and Address for Formulator........................................
|
| 3. |
Common Name of the active ingridient(s).........................................
|
| 4. |
Concentration of active ingridients(s)...............................................
|
| 5. |
Biological source of a.i. (for botanical specify the plant part, stage of growth etc.)
|
| 6. |
Name & Location of producer of a.i...................................
|
| 7. |
Chemical Name...................................................................
|
| 8. |
Formulation type ..........................................................
|
| 9. |
Proposed Uses.................................................................
|
| 10. |
packaging/Containers (Material, size)................................................
|
| 11. |
Registrant (Name, Address, Status)...........................................
|
| 12. |
Agents/Distributors in Kenya..............................................
|
| 13. |
Premises (Reg.No. Date of issue)................................................
CHEMISTRY DATA
|
| 14. |
Physical/Chemical Properties of the a.i ...............................................
| (b) |
Physical/Chemical properties of the technical grade material....................................
|
| (c) |
Composition of the technical product (purity %, natures & identity of impurities other details should be provided in the dossier)...........................................
|
| (d) |
Physical/Chemical Properties of the Formulated Product..........................................
|
| (e) |
Composition of the Formulated Product (Concentration of a.i in the formulation. other details should be provided in the dossier)...................................................
|
| (f) |
Method of analysis for determination of the a.i in technical and formulated products (State all the methods for different components)
|
...............................................
...............................................
Biological(efficacy) Data
| (a) |
Target Pest(s), Diseases(s), Host(s).....................................................
|
| (c) |
Method, Rate, Frequency of application...................................................
|
| (d) |
Recommendation for use in Kenya...........................................................
|
| (e) |
Recommendations for use by authorized bodies outside Kenya..............................................
|
| (a) |
Acute Toxicological Data of the active ingridient(s)..............................................
|
| (b) |
Acute toxicity data of the formulated product:-..................................................
|
| (c) |
Short term toxicity studies..................................................................
|
| (d) |
Other toxicological studies:
|
|
| (1) |
Reproduction studies...................................................................
|
| (2) |
Teratological studies...................................................................
|
| (3) |
Neurotoxicity studies.....................................................................
|
| (4) |
Mutagenecity studies...............................................................
|
| (5) |
Long term toxicity/carcinogenicity studies...................................
|
| (6) |
Accumulation of compound in tissues............................................
|
| (7) |
Metabolic studies.........................................................
|
| (8) |
Effects on livestock, poultry.............................................
|
| (9) |
Toxicity Data on Impurities......................................................
|
| (10) |
Toxicity Data on metabolites...............................................
|
| (11) |
Human toxicology and medical aspects:
|
| (1) |
Hazards to humans.............................................................
|
| (2) |
Symptomps of poisoning.....................................................
|
| (3) |
Antidote...........................................................
|
| (4) |
Treatment...........................................................................................
|
| (5) |
First Aid Measures...................................................................................
|
| (6) |
Safety Precautions/Restrictions........................................................................
| (a) |
Principal Residues............................................................................
|
| (b) |
Disappearance and fate of residues...........................................................
|
| (c) |
Method(s) of analysis (crops, soil, water, feedstuffs etc.).......................................
|
| (a) |
Degradation and mobility studies (Soil, water, air)..............................................
|
| (b) |
Toxicity to birds......................................................
|
| (c) |
Toxicity to fish.......................................................
|
| (d) |
Toxicity to honeybees/beneficial insects.....................................
|
| (e) |
Toxicity to earthworms, other soil invertebrates........................................
|
| (f) |
Changes in soil ecology.......................................................
|
Information on Approvals/Registrations in other countries..................................................
Draft of local label (as per Legal Notice No. 89/1984).
............................................................................................
Brief prepared by............................................................
Signature.....................................................................
Official stamp......................................................................
Date...................................................................
Decision of the PCPB registration Sub-Committee
Recommended/Not Recommended for registration..
Reasons:-
..............................................................................................
..............................................................................................
Date......................................................................................
|
_____________________________
|
FORM B4
|
(r. 4H)
|
|
P.O BOX 13794-00800, NAIROBI, WAIYAKI WAYTel: 254-020 444611/4450242 Fax: 254-020 4449072E-MAIL: pcpboard@todays.co.keWEBSITE ADDRESS: www.pcpb.or.ke
|
|
|
SUMMARY OF THE DATA SUBMITTED TO THE PCPB FOR REGISTRATION OF A SEMIOCHEMICAL PEST CONTROL PRODUCT
| 1. |
Trade Name...................................................................
|
| 2. |
The Name and Address of Formulator.................................................
|
| 3. |
Common Name of the active ingredient(s)............................................
|
| 4. |
Concentration of active ingredient(s)...............................................
|
| 5. |
Source of a.i (natural or synthetic)......................................................
|
| 6. |
Name & Location of producer of a.i....................................................
|
| 7. |
Chemical Name...........................................................
|
| 8a. |
Formulation type.........................................................
|
| 8b. |
Associated Device..........................................................
|
| 9. |
Proposed Uses.................................................................
|
| 10. |
Packaging/Containers (Material, size).............................................
|
| 11. |
Registrant (Name, Address,)..........................................................
|
| 12. |
Agent/Distributors in Kenya........................................................
|
| 13. |
Premises (Registration No. Date of issue).................................................
CHEMISTRY DATA
|
| 14(a) |
Physical/Chemical propoerties of the technical grade material............................
|
| (b) |
Composition of the tecnical product (purity %, nature & identity of impurities other details should be provided in the dossier).......................................
|
| (c) |
Physical/Chemical Properties of the Formulated Product....................................
|
| (d) |
Composition of the Formulated Product (Concentration of a.i in the formulation, Other details should be provided in the dossier).................................................
|
| (e) |
Method of analysis for determination of the a.i in technical and formulated products (State all the methods for different components)................................................
TOXICOLOGICAL DATA
(A) Technical grade active ingridient(s)
TIER I Requirements
| (a) |
Acute Toxicological Data of the Technical grade active ingridient(s) Straight-Chain Lepidopteran Pheromones (SCLPs) are exempt from all toxicological data requirements. The following studies are required for non-SCLPs
|
Acute oral LD50 ....................................................................................
Acute dermal LD50.....................................................................................
Inhalation LC50...................................................................................
| (b) |
Short term toxicity studies.....................................................................
|
| (c) |
Mutagenecity studies.........................................................................
|
TIER II Requirements (Information is required are triggered by TIER I studies
|
| (1) |
Reproduction studies.........................................................................
|
| (2) |
Teratological studies........................................................................
|
| (3) |
Neurotoxicity studies...........................................................................
|
| (4) |
Additional mutagenecity studies................................................................
|
| (5) |
Carcinogenicty studies..........................................................................
|
| (6) |
Chronic toxicity................................................................................
|
| (7) |
Hypersensitivity/allergies in human or any other human exposure data ............................
|
| (8) |
Metabolic studies................................................................................
(B) Acute toxicity data of the formulated product:
SCLPs are exempt provided the co-formulants are not of toxicological concern (MSDS must be provided.) The Acute toxicity studies will be provided for non-SCLPs if any of the co-formulants are of toxicological concern.
.................................................................................................
.................................................................................................
|
EMERGENCY PROCEDURE IN CASE OF A ACCIDENTAL EXPLOSURE OR POISONING
| (a) |
Hazards to humans........................................................................
|
| (b) |
Symptomps of poisoning ....................................................................
|
| (c) |
Antidote..................................................................................
|
| (d) |
Treatment................................................................................
|
| (e) |
First Aid Measures.........................................................................
|
| (f) |
safety Precautions/Restrictions............................................................
|
| (a) |
Toxicity to birds (Reguired if the product could be ingested by birds, e.g. a granular formulation) ....................
|
| (b) |
Toxicity to fish (Required if product is applied by air, or directly to water or at a rate exceeding natural background levels) ..............................................
|
| (c) |
Freshwater invertebrates (Required if product is applied by aircraft, or directly to water or at a rate exceeding natural background levels) ...........................................................
|
| (d) |
Algae (Waived for products in affixed dispensers and if exposure is unlikely to exceed natural background levels) ..............
|
| (e) |
Toxicty to bees (Information/discussion, to address whether behaviour or reproduct would be affected, is required if exposure is likely to exceed natural background levels) ...................................................................
|
| (f) |
Toxicity to earthworms (Required if product is applied to soil and can accumulate in soil. Required if exposure exceeds natural background levels) ....................................................................
|
Information on Approvals/Registrations in other countries........................................................
Draft of local label (as per Legal Notice No. 89/1984).
................................................................................................................
Brief prepared by...........................................................
Signature..................................................................
Official stamp..............................................................
Date.........................................................................
|
PART X (For official use only)
|
|
Decision of the PCPB registration Sub-Committee
Recommended/Not Recommended for registration
Reasons:-
Date................................................
_______________________________
|
PEST CONTROL PRODUCTS ACT[Cap. 346]PEST CONTROL PRODUCTS (REGISTRATION) REGULATIONS, 2006
|
|
APPLICATION FOR THE INTRODUCTION OF NEW PEST CONTROL PRODUCT
(To be completed and submitted in triplicate)
|
To:
|
The Managing DirectorPest Control Products BoardP.O. Box 13794-00800Westlands, Nairobi
|
APPLICANT'S NAME AND ADDRESS.........................................................................
Tel No. ............................. Fax No. ..............................................................
STATUS OF APPLICANT (Manufacturer, agent etc.)...................................................................
| 1. |
Approved Common Name(s)......................................................
|
| 2. |
Chemical Name...................................................................................
|
| 3. |
Chemical formula.......................................................................................
|
| 4. |
Chemical structure.....................................................................................
|
| 5. |
Trade Name(s).........................................................................................
|
| 6: |
Proposed Kenyan Name(s)........................................................................
|
| 7: |
Formulation Type (W.P.E.C., Dust etc.).............................................................
|
| 8. |
Concentration of Each Active Ingiredients...............................................................
|
| 9. |
Quantity required for testing...........................................................................
|
| 10. |
Proposed Uses (Agricultural, Health, Veterinary, Forestry etc.)..........................................
|
| 11. |
Location and Area of Test Plots......................................................................
|
| 12. |
Target pest(s) Host(s) or Area of Application.................................................................
|
| 13. |
Mode of action.............................................................................................
|
| 14. |
Toxicity of the product to test animals (Acute Oral and Dermal LD50 Inhalition LC50 etc.).....................
|
| 15. |
The effects of the product on the environment-
| (a) |
Toxicity to bees....................................................
|
| (b) |
Toxicity to fish....................................................
|
| (c) |
Toxicity to birds...................................................
|
| (d) |
Toxicity to soil micro-organism........................................
|
|
| 16. |
Proposed precautions to users.......................................
|
| 17. |
Antidote, Treatment of poisoning................................................
|
| 18. |
Shelf life of the product..............................................
|
| 19. |
Country of Origin of the product..................................................
|
| 20. |
Name and Address of manufacturer.....................................................
|
| 21. |
Name and address of formulator........................................................
|
| 22. |
Countries where tested and registered ..................................................
|
| 23. |
Ownership of data (name, address)......................................................
|
| 24. |
Patent sit (lilt ion/patent holder)......................... Confirm that the information contained herein is true to the best of my knowledge and belief.
|
................................................
|
........................................
|
|
Date of application
|
Signature of applicant
|
Name........................... Designation/Position held ..............................
Note
Every applicant must be accompanied by
|
| 1. |
Supporting date and information which should include -
| (a) |
Chemistry specifications, composition of the product, and the technical a.i method of analysis of the a.i determination
|
| (b) |
information on biological activity on the product, directions for use
|
| (c) |
Metablisim, Residues, methods of analysis for residues
|
| (d) |
Toxicological data on the technical and formulated product(s)
|
|
| 2. |
Experimental labels (typed)
|
| 3. |
Analytical standards (approximate 100% a.i) 1.0 gram.
|
_____________________________
|
PEST CONTROL PRODUCTS ACT
|
|
[Cap. 346]
|
PEST CONTROL PRODUCTS (REGISTRATION) REGULATION, 2006
|
Date ........................................
|
|
|
|
REF: PERMIT NO. ...............................
|
PERMIT FOR EXPERIMENTAL AND EFFICACY TRIALS OF NEW PEST CONTROL PRODUCTS
This is to grant permission as requested for your Centre/Organization to carry out efficacy trials of the new pest control product(s) as indicated below -
|
Pest Control Product(s)
|
Crop(s)/Commodity(ies)/Use(s)/Target Pest(s)
|
You are requested to inform the Pest Control Products Board of the commencement of the experimental/efficacy trials and also periodically submit to the Board progress reports. The trial should be carried out using a Pest Control Products Board approved trial protocol. At the conclusion of the experimental/efficacy trial, a detailed confidential report on the performance of the candidate pesticide and recommendations for its use shall be submitted to the Board quoting the above reference and date.
It would be highly appreciated if trials are compeleted as quickly as possible to avoid delays in introducing suitab;le products in the market. The company will provide you with the required trial samples/materials but the Board shall not meet expenses for the trials.
It is the responsibility of the applicant to ensure that the efficacy trials are carried out to the satisfaction of the Board.
Managing Director
Pest Control Products Board
_____________________________
CHANGE OF SOURCE
PRODUCT INFORMATION
| 1. |
Trade Name:.................................................
|
| 2. |
Common name of active ingridient(s) and Concentration:...........................................
|
| 3. |
PCPB registration No:...................................
|
| 4. |
Technical Grade Active ingridient
| (a) |
Name of basis manufacture:..........................................
|
| (b) |
Physical Location of basic manufacture:...............................
|
| (i) |
Postal Address: ...................................................
|
| (ii) |
Telephone No: .................................................
|
| (iii) |
E-Mail: ...............................................
|
| (iv) |
Fax No: ...........................................
|
| (v) |
Street/Road: ............................................
|
| (d) |
Specifications (with analytical proof)
|
| (e) |
Relationship with Registrant, if different
|
|
| 5. |
| (a) |
Name of Formulator: ................................
|
| (b) |
Physical Location: ....................................
|
| (c) |
| (i) |
Postal Address:................................. |
| (ii) |
Telephone No: ........................................ |
| (iii) |
Fax No: ...................................... |
| (iv) |
E-Mail: ....................................... |
| (v) |
Street/Road: ........................................... |
|
| (d) |
Composition (with analytical proof)
|
|
| 6. |
Name of new source ..........................................
|
| 7. |
Relationship with old source/manufacturer/formulator, if more than one.
|
THE PEST CONTROL PRODUCTS (LABELLING, ADVERTISING AND PACKAGING) REGULATIONS
ARRANGEMENT OF REGULATIONS
| 3. |
Labelling requirements
|
| 5. |
Notice on display panel
|
| 6. |
Display of additional information
|
| 7. |
Information where product is distributed in bulk
|
SCHEDULES
| FIRST SCHEDULE [r. 3(2)] — |
CLASSIFICATION OF PEST CONTROL PRODUCTS
|
| SECOND SCHEDULE [r. 3(2)] — |
CAUTIONARY SYMBOLS AND WORDS
|
THE PEST CONTROL PRODUCTS (IMPORTATION AND EXPORTATION) REGULATIONS, 1984
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Importation and Exportation) Regulations, 1984.
|
| 2. |
Importation and exportation prohibited unless licensed
| (1) |
No person shall import or export a pest control product for commercial purposes unless that person is in possession of a licence issued under these Regulations.
|
| (2) |
No licence shall be issued for the importation of a pest control product unless the pest control product is registered in accordance with the Pest Control Products (Registration) Regulations, 1984.
|
| (3) |
No licence shall be issued under these Regulations unless the Board is satisfied that the provisions of the Pest Control Products Act, 1982 have been complied with.
|
|
| 3. |
Presumptions
| (1) |
For the purpose of paragraph (1) of regulation 2, a pest control product shall be considered to be for commercial purposes if it is for a use other than scientific research or testing and products imported for resale, manufacture, formulation, owner’s use or commodity aid shall be deemed to be imported for commercial purposes.
|
| (2) |
For the purpose of paragraph (1) of regulation 2, a pest control product shall be deemed to have been exported when it is placed on any ship, aircraft, train or vehicle within Kenya for the purpose of export.
|
|
| 4. |
Application for licences
| (1) |
Any person desiring a licence in respect of importation or exportation of a pest control product for commercial purposes shall apply to the Board for a licence in Form PCPB 1 set out in the Schedule which form shall be signed by the importer or exporter, as the case may be, who shall state the purpose of the importation or exportation of the pest control product in the following manner—
| (i) |
"FOR RESALE", where the product is being imported or exported for the purpose of resale whether in the original pack or after repacking; or |
| (ii) |
"FOR MANUFACTURING PURPOSES", where the pest control product is being imported or exported for use in the manufacture or formulation of a registered pest control product; or |
| (iii) |
"FOR IMPORTER’S OWN USE", where the pest control product is being imported into Kenya or exported to another country for the sole use of the importer, together with information respecting where the pest control product shall be used and the nature of that use. |
|
| (2) |
The Board shall from time to time determine the fee to be paid in respect of an application for a licence.
|
|
| 5. |
Dealing with applications and issue of licences, etc.
| (1) |
The Board may approve an application made under regulation 4 if it is satisfied that—
| (a) |
the application contains the information required under that regulation;
|
| (b) |
the pest control product contains ingredients that have been previously assessed or evaluated for the purposes of the Act and these Regulations and which have been accepted for registration under the Pest Control Products (Registration) Regulations, 1984, for the use stated in the application;
|
| (c) |
premises where the pest control product is to be stored, processed, warehoused, used, repacked or sold shall have been duly licensed by the Board,
|
and issue a licence in Form PCPB 1 set out in the Schedule.
|
| (2) |
Where the Board does not approve an application it shall not be obliged to give any reason therefor.
|
| (3) |
No person to whom a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence to any other person without the approval of the Board, which approval shall be endorsed on the licence.
|
| (4) |
The Board may attach to a licence such conditions as it deems fit to impose and in particular any such condition may restrict the importation and exportation of a pest control product from a specified source or for specified usage and the Board may, from time to time, vary, add to or revoke any of the conditions attached to a licence.
|
| (5) |
The licence issued under regulation 2(1) shall be valid for three (3) months from the date of issue.
[L.N. 125/2006, r. 2.]
|
|
| 6. |
Duty of licensees, etc.
| (1) |
Repealed by L.N. 125/2006, r. 3.
|
| (2) |
It shall be the sole responsibility of the importer or his agent to ensure that Form PCPB 1 is endorsed by the Customs Department at the port of entry and to forward a signed copy to the Board
[L.N. 125/2006, r. 3.]
|
|
| 7. |
Duty of customs officers
Where for any reason a customs officer at a port of entry withholds the release of any pest control product he shall forthwith advise the Board of that action.
|
| 8. |
Cancellation and suspension of licences
| (1) |
The Board may cancel or, for such period as it thinks fit, suspend a licence issued under these Regulations if the Board is satisfied that—
| (a) |
the licensee or his servant or agent has been convicted of an offence under the Act; or
|
| (b) |
the licensee or his servant or agent has committed a breach of any of the terms or conditions of the licence; or
|
| (c) |
for any other reason, it is in the public interest so to do.
|
|
| (2) |
Before cancelling or suspending a licence under this Regulation the Board shall give the licensee twenty-eight days’ notice to show cause why the licence should not be cancelled or suspended and the Board’s decision in the matter shall be final.
|
|
| 9. |
Imported pest control products to comply with these Regulations
No person shall package, store, transport, display, distribute, sell or otherwise deal in a pest control product that has not been imported in accordance with these Regulations.
[L.N. 125/2006, r. 4.]
|
| 10. |
Licence prescribed form
A licence to import or export a pest control product under these Regulations shall be in the prescribed Form PCPB2 set out in the Schedule.
[L.N. 125/2006, r. 5.]
|
THE PEST CONTROL PRODUCTS (LABELLING, ADVERTISING AND PACKAGING) REGULATIONS, 1984
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Labelling, Advertising and Packaging) Regulations, 1984.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"display panel" means part of a label applied on or affixed to the package of a pest control product but does not include a leaflet or brochure unless it is part of the label;
"Formulator" means a person who prepares the active ingredient of a pest control product into a form which is convenient for its intended use;
"Re-packer" means a person who puts a formulated product into appropriate unit sizes.
[L.N. 127/2006, r. 2.]
|
| 3. |
Labelling requirements
| (1) |
No pest control product shall be distributed or sold without a label.
|
| (2) |
No label shall be used on a pest control product unless it has been approved by the Board and, unless the Board otherwise directs, every label shall show—
| (a) |
the name of a pest control product which shall be descriptive of the physical form and the purpose of the pest control product and shall include the common name of its active ingredients and may include a distinctive brand or trade mark;
|
| (b) |
the class designation of the pest control product in capital letters and shall be classified as set out in the First Schedule;
|
| (c) |
information detailing the nature and degree of hazard inherent in the pest control product and the nature and degree of hazard shall be identified by the appropriate precautionary symbol and signal words selected from the Second Schedule together with a statement respecting the nature of the primary hazard to which the symbol relates;
|
| (d) |
a statement directing the user to read the label which statement shall be in the following form – "READ THE LABEL BEFORE USING";
|
| (e) |
a guarantee statement in the following manner—
| (i) |
the word in capital letters "GUARANTEE", followed by; |
| (ii) |
a colon; followed by; |
| (iii) |
the common name of the active ingredient of pest control product or where a common name has not been designated, the chemical or other name of the active ingredient; followed by; |
| (iv) |
the contents of the active ingredient expressed— |
|
| (f) |
the registration number of the pest control product which shall be set out in the following manner—
"REGISTRATION NO. PCPB (CR) 0000" AND PCPB (TCR) 0000";
Where "CR" means Certificate of Registration for a period of three years and or renewal for a period not exceeding two years at any one time; and "TCR" means Temporary Certificate of Registration for a period not exceeding one year;
|
| (g) |
a statement of the net contents of the package for the pest control product, which shall be expressed—
| (i) |
in the case of a liquid pest control product of a volume less than one litre, in terms of millilitres; |
| (ii) |
in the case of a liquid pest control product of a volume of one litre or more, in terms of litres; |
| (iii) |
in the case of a pressure-packed pest control product in terms of weight, in grams if less than one kilogram; |
| (iv) |
in the case of a pressure-packed pest control product which weighs one kilogram or more, in terms of kilograms; |
| (v) |
in the case of a dry formulation pest control product which weighs less than a kilogram, in terms of grams; |
| (vi) |
in the case of a dry formulation pest control product which weighs one kilogram or more, in terms of kilograms; |
| (vii) |
in the case of a pest control product that is semi-solid or viscous in terms of either weight or volume in accordance with this paragraph; or |
| (viii) |
in the case of a pest control product the nature of which does not lend itself to net content statements prescribed in this paragraph, in terms acceptable to the Board; |
|
| (h) |
the name and postal address of the registrant and the name and postal address of the resident agent, if any and the names and postal addresses of manufacturer, formulator, distributor and re-packer;
|
| (i) |
the directions for use of the pest control product, which directions shall include dosage rates, timing of application and use limitations;
|
| (j) |
information identifying any significant hazards respecting the handling, storage, display, distribution and disposal of the pest control product which information shall include instructions respecting procedures to alleviate the hazard and when required by the Board, instructions respecting decontamination procedures and disposal of the pest control product and the empty package;
|
| (k) |
information identifying any significant hazard to—
| (i) |
things on or in relation to which the pest control product is intended to be used; or |
| (ii) |
public health, plants, animals or the environment, which information shall include instructions respecting the procedures to alleviate any such hazard; |
|
| (l) |
instructions for first aid, which instructions shall be under the heading in capital letters "FIRST AID INSTRUCTIONS" and shall set out the practical measures to be taken in the event of poisoning or other injury caused by the pest control product;
|
| (m) |
the toxicological information essential to the treatment of a person poisoned or otherwise injured by the pest control product, which information shall be under the heading in capital letters "TOXICOLOGICAL INFORMATION" and shall—
| (i) |
describe the symptoms of poisoning; |
| (ii) |
state antidotes and remedial measures; and |
| (iii) |
state the ingredient that may affect the treatment; |
|
| (n) |
a notice to the user of the pest control product which notice shall be in the following manner;
|
|
This pest control product is to be used only in accordance with the directions on this label. It is an offence under the Pest Control Products Act (Cap. 346) to use or store a pest control product under unsafe conditions;
| (o) |
the colour codes banding precautionary pictograms relating to the toxicity of the product in the following manner of declining toxicity—
| (i) |
PMS Red 199C for products classified under WHO class Ia and Ib; |
| (ii) |
PMS yellow C for products classified under WHO class II; |
| (iii) |
PMS Blue 293C for products classified under WHO class III; |
| (iv) |
PMS Green 347C for unclassified products under WHO classification; |
| (v) |
the colour shall not be used elsewhere on the label; and |
| (vi) |
any other pictogram as directed by the Board; |
|
| (p) |
separate pictograms depicting at least one pest controlled by the pest control product and crop or animal or other usage as registered under this Act;
|
| (q) |
an instruction directing the user on storage which shall be in capital letters– KEEP LOCKED OUT OF REACH OF CHILDREN;
|
| (r) |
information on the date of manufacture, expiry date, batch number and shelf-life if stored in original container under cool dry conditions.
|
| (3) |
The label for a pest control product which is a device of a type or kind listed in the First Schedule to the Pest Control Products (Registration) Regulations shall contain the information referred to in subparagraphs (f), (h), (i), (j), and (k) of paragraph (2).
[L.N. 127/2006, r. 3.]
|
|
| 4. |
Display panels
| (1) |
The display panel shall consist of one principal display panel and at least one secondary display panel.
|
| (2) |
Where the primary purpose of a pest control product is not to control, prevent, destroy, mitigate, attract or repel a pest, but it has these properties, the pest control product shall have a display panel with—
| (a) |
the information referred to in subparagraphs (a), (b), (c), (d), (g) and (h) of regulation 3(2) on the principal display panel; and
|
| (b) |
the information referred to in subparagraphs (e), (f), (i), (l) and (m) of regulation 3(2) on the secondary display panel.
|
|
| (3) |
Where the primary purpose of a pest control product is to control, prevent, destroy, mitigate, attract or repel a pest, the pest control product shall have a display panel with the information referred to in subparagraphs (a), (b), (c), (d), (e), (f), (g) and (h) of regulation 3(2) on the principal display panel.
|
| (4) |
This Regulation shall apply to a label for a pest control product that is within the meaning of paragraph (a) of the definition of "pest control product" in section 2 of the Act.
|
|
| 5. |
Notice on display panel
| (1) |
Where the principal display panel shows the pest control product as "RESTRICTED", the notice referred to in regulation 3(2)(n) shall appear prominently at the top of the secondary display panel followed by the heading in capital letters "RESTRICTED USE", followed by the directions for use, dosage, timing of application and use limitations to which the restrictions relates all of which shall be circumscribed by a line to set the information apart from all other information required to be shown on the secondary display panel.
|
| (2) |
Notwithstanding paragraph (1), where the principle display panel shows the pest control product as "RESTRICTED" the directions for use, dosage rates, timing of application and use limitations to which the restriction relates, together with the information referred to in subparagraphs (a), (b), (c), (d), (e), (f), (g), (h) and (i) of regulation 3(2) may, with the approval of the Board, appear in a brochure or leaflet that will accompany the package for the pest control product.
|
| (3) |
Where the information required to be shown on the label is not included in the display panel, the display panel shall contain the words in capital letters "READ ATTACHED BROCHURE BEFORE USE" prominently displayed thereon.
|
|
| 6. |
Display of additional information
| (1) |
Subject to the approval of the Board, additional information relating to the pest control product and any graphic design or symbol maybe shown on the label if it does not unreasonably detract or obscure the information required to be shown on the label under these Regulations.
|
| (2) |
A registrant may include on the label the following liability limitation warranty—
Seller’s guarantee is limited to the terms set out on the label and subject thereto, the buyer assumes the risk to persons or property arising from the use or handling of this product and accepts the product on that condition.
|
|
| 7. |
Information where product is distributed in bulk
Where a pest control product is distributed in a bulk container, the information referred to in subparagraphs (a), (b), (e), (f), (g), (h), (i) and (m) of regulation 3(2) shall be shown—
| (a) |
on the bulk container; and
|
| (b) |
on the documents respecting the pest control product or on a statement accompanying the consignment.
|
|
| 8. |
Information on label
| (1) |
The information on every label shall be printed in both English and Kiswahili languages.
|
| (2) |
All units of measures shown on the label shall be expressed in accordance with the requirements of the Weights and Measures Act (Cap. 513).
|
| (3) |
All information shown on the label shall be printed in a manner that is conspicuous, legible and indelible.
|
|
| 9. |
Denaturation
Where the physical properties of a pest control product are such that the presence of the pest control product may not be recognized when it is used, and is likely to expose a person or domestic animal to a severe health risk, the pest control product shall be denatured by means of colour, odour or such other means as the Board may approve to provide signal or warning of its presence.
|
| 10. |
Storage and display
A pest control product shall be stored and displayed in accordance with the conditions shown on the label.
|
| 11. |
Distribution
Notwithstanding the provisions of the Pharmacy and Poisons Act (Cap. 244), a pest control product shall be distributed in accordance with such condition as the Board shall specify.
|
| 12. |
Prohibition
No person shall use a pest control product in a manner that is inconsistent with the directions or limitations respecting its use as shown on the label.
|
| 13. |
Packaging
| (1) |
Every package for a pest control product shall, unless the Board otherwise directs, be approved by the Board.
|
| (2) |
The package for every pest control product shall be sufficiently durable and be designed and manufactured to contain the pest control product safety under practical conditions of storage, display and distribution.
|
| (3) |
Every package shall be designed and manufactured to permit—
| (a) |
the withdrawal of any or all of the contents in a manner that is safe to the user; and
|
| (b) |
the closing of the package in a manner that will contain the pest control product satisfactorily under practical conditions.
|
|
| (4) |
Every package shall be constructed in such a manner as to minimize the degradation or change of its contents resulting from interaction or from the effects of radiation or other means.
|
|
| 14. |
General prohibitions
| (1) |
A label shall not contain any information respecting any organism or causative agent of a disease that is required to be reported under the Animal Diseases Act (Cap. 364).
|
| (2) |
Information that is required under these Regulations to be shown on a label shall not appear at the bottom of the package.
|
| (3) |
Words stating, implying or inferring that a pest control product is approved, accepted or recommended by the government or by any department or agency thereof shall not appear on a package or label or in any advertisement respecting a pest control product.
|
|
FIRST SCHEDULE
[Regulation 3(2), L.N. 127/2006, ss. 4, 5 and 6.]
CLASSIFICATION OF PEST CONTROL PRODUCTS
| 1. |
Restricted Class
Where the Board has set forth additional essential conditions to be shown on the label respecting the display, distribution, use, limitations or qualifications of persons who may use the pest control product, the pest control product shall have the following properties—
| (a) |
acute oral LD50 is greater than 50 mg/kg body weight;
|
| (b) |
acute dermal LD50 is greater than 100 mg/kg body weight;
|
| (c) |
environmental risks are significant and will be judged accordingly;
|
| (d) |
pest control products used in aquatic and forestry situations are classified restricted.
|
[L.N. 127/2006, rr. 4 and 5.]
|
| 2. |
Commercial and Agricultural Class
Where the pest control product is to be displayed and distributed for general use in commercial activities specified on the label it shall have the following properties—
| (a) |
acute oral LD50 is greater than 50 mg/kg body weight;
|
| (b) |
acute dermal LD50 is greater than 100 mg/kg body weight;
|
| (c) |
environmental effects possible in limited regions.
|
|
| 3. |
Domestic Class
Where the pest control product is to be displayed and distributed for the use in and around a dwelling it shall have the following properties—
| (a) |
acute oral LD50 is over 500 mg/kg body weight;
|
| (b) |
acute dermal LD50 is over 1,000 mg/kg body weight;
|
| (c) |
no special precautions or equipment required for inhalation hazard;
|
| (d) |
no irreversible effects from repeated exposures;
|
| (e) |
disposal of product and containers can safely be done by placing in garbage;
|
| (f) |
package sizes limited to amounts that can be safely used and stored by consumers.
|
|
SECOND SCHEDULE
CAUTIONARY SYMBOLS AND WORDS
THE PEST CONTROL PRODUCTS (LABELLING, ADVERTISING AND PACKAGING) REGULATIONS
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Labelling, Advertising and Packaging) Regulations.
|
| 2. |
Interpretation
In these Regulations, unless the context otherwise requires—
"display panel" means part of a label applied on or affixed to the package of a pest control product but does not include a leaflet or brochure unless it is part of the label;
"Formulator" means a person who prepares the active ingredient of a pest control product into a form which is convenient for its intended use;
"Re-packer" means a person who puts a formulated product into appropriate unit sizes.
[L.N. 127/2006, r. 2.]
|
| 3. |
Labelling requirements
| (1) |
No pest control product shall be distributed or sold without a label.
|
| (2) |
No label shall be used on a pest control product unless it has been approved by the Board and, unless the Board otherwise directs, every label shall show—
| (a) |
the name of a pest control product which shall be descriptive of the physical form and the purpose of the pest control product and shall include the common name of its active ingredients and may include a distinctive brand or trade mark;
|
| (b) |
the class designation of the pest control product in capital letters and shall be classified as set out in the First Schedule;
|
| (c) |
information detailing the nature and degree of hazard inherent in the pest control product and the nature and degree of hazard shall be identified by the appropriate precautionary symbol and signal words selected from the Second Schedule together with a statement respecting the nature of the primary hazard to which the symbol relates;
|
| (d) |
a statement directing the user to read the label which statement shall be in the following form – "READ THE LABEL BEFORE USING";
|
| (e) |
a guarantee statement in the following manner—
| (i) |
the word in capital letters "GUARANTEE", followed by; |
| (ii) |
a colon; followed by — |
| (iii) |
the common name of the active ingredient of pest control product or where a common name has not been designated, the chemical or other name of the active ingredient; followed by; |
| (iv) |
the contents of the active ingredient expressed— |
|
| (f) |
the registration number of the pest control product which shall be set out in the following manner—
"REGISTRATION NO. PCPB (CR) 0000" AND PCPB (TCR) 0000";
Where "CR" means Certificate of Registration for a period of three years and or renewal for a period not exceeding two years at any one time; and "TCR" means Temporary Certificate of Registration for a period not exceeding one year;
|
| (g) |
a statement of the net contents of the package for the pest control product, which shall be expressed—
| (i) |
in the case of a liquid pest control product of a volume less than one litre, in terms of millilitres; |
| (ii) |
in the case of a liquid pest control product of a volume of one litre or more, in terms of litres; |
| (iii) |
in the case of a pressure-packed pest control product in terms of weight, in grams if less than one kilogram; |
| (iv) |
in the case of a pressure-packed pest control product which weighs one kilogram or more, in terms of kilograms; |
| (v) |
in the case of a dry formulation pest control product which weighs less than a kilogram, in terms of grams; |
| (vi) |
in the case of a dry formulation pest control product which weighs one kilogram or more, in terms of kilograms; |
| (vii) |
in the case of a pest control product that is semi-solid or viscous in terms of either weight or volume in accordance with this paragraph; or |
| (viii) |
in the case of a pest control product the nature of which does not lend itself to net content statements prescribed in this paragraph, in terms acceptable to the Board; |
|
| (h) |
the name and postal address of the registrant and the name and postal address of the resident agent, if any and the names and postal addresses of manufacturer, formulator, distributor and re-packer;
|
| (i) |
the directions for use of the pest control product, which directions shall include dosage rates, timing of application and use limitations;
|
| (j) |
information identifying any significant hazards respecting the handling, storage, display, distribution and disposal of the pest control product which information shall include instructions respecting procedures to alleviate the hazard and when required by the Board, instructions respecting decontamination procedures and disposal of the pest control product and the empty package;
|
| (k) |
information identifying any significant hazard to—
| (i) |
things on or in relation to which the pest control product is intended to be used; or |
| (ii) |
public health, plants, animals or the environment, which information shall include instructions respecting the procedures to alleviate any such hazard; |
|
| (l) |
instructions for first aid, which instructions shall be under the heading in capital letters "FIRST AID INSTRUCTIONS" and shall set out the practical measures to be taken in the event of poisoning or other injury caused by the pest control product;
|
| (m) |
the toxicological information essential to the treatment of a person poisoned or otherwise injured by the pest control product, which information shall be under the heading in capital letters "TOXICOLOGICAL INFORMATION" and shall—
| (i) |
describe the symptoms of poisoning; |
| (ii) |
state antidotes and remedial measures; and |
| (iii) |
state the ingredient that may affect the treatment; |
|
| (n) |
a notice to the user of the pest control product which notice shall be in the following manner;
NOTICE TO USER
This pest control product is to be used only in accordance with the directions on this label. It is an offence under the Pest Control Products Act (Cap. 346) to use or store a pest control product under unsafe conditions;
|
| (o) |
the colour codes banding precautionary pictograms relating to the toxicity of the product in the following manner of declining toxicity—
| (i) |
PMS Red 199C for products classified under WHO class Ia and Ib; |
| (ii) |
PMS yellow C for products classified under WHO class II; |
| (iii) |
PMS Blue 293C for products classified under WHO class III; |
| (iv) |
PMS Green 347C for unclassified products under WHO classification; |
| (v) |
the colour shall not be used elsewhere on the label; and |
| (vi) |
any other pictogram as directed by the Board; |
|
| (p) |
separate pictograms depicting at least one pest controlled by the pest control product and crop or animal or other usage as registered under this Act;
|
| (q) |
an instruction directing the user on storage which shall be in capital letters– KEEP LOCKED OUT OF REACH OF CHILDREN;
|
| (r) |
information on the date of manufacture, expiry date, batch number and shelf-life if stored in original container under cool dry conditions.
|
|
| (3) |
The label for a pest control product which is a device of a type or kind listed in the First Schedule to the Pest Control Products (Registration) Regulations shall contain the information referred to in subparagraphs (f), (h), (i), (j), and (k) of paragraph (2).
[L.N. 127/2006, r. 3.]
|
|
| 4. |
Display panels
| (1) |
The display panel shall consist of one principal display panel and at least one secondary display panel.
|
| (2) |
Where the primary purpose of a pest control product is not to control, prevent, destroy, mitigate, attract or repel a pest, but it has these properties, the pest control product shall have a display panel with—
| (a) |
the information referred to in subparagraphs (a), (b), (c), (d), (g) and (h) of regulation 3(2) on the principal display panel; and
|
| (b) |
the information referred to in subparagraphs (e), (f), (i), (l) and (m) of regulation 3(2) on the secondary display panel.
|
|
| (3) |
Where the primary purpose of a pest control product is to control, prevent, destroy, mitigate, attract or repel a pest, the pest control product shall have a display panel with the information referred to in subparagraphs (a), (b), (c), (d), (e), (f), (g) and (h) of regulation 3(2) on the principal display panel.
|
| (4) |
This Regulation shall apply to a label for a pest control product that is within the meaning of paragraph (a) of the definition of "pest control product" in section 2 of the Act.
|
|
| 5. |
Notice on display panel
| (1) |
Where the principal display panel shows the pest control product as "RESTRICTED", the notice referred to in regulation 3(2)(n) shall appear prominently at the top of the secondary display panel followed by the heading in capital letters "RESTRICTED USE", followed by the directions for use, dosage, timing of application and use limitations to which the restrictions relates all of which shall be circumscribed by a line to set the information apart from all other information required to be shown on the secondary display panel.
|
| (2) |
Notwithstanding paragraph (1), where the principle display panel shows the pest control product as "RESTRICTED" the directions for use, dosage rates, timing of application and use limitations to which the restriction relates, together with the information referred to in subparagraphs (a), (b), (c), (d), (e), (f), (g), (h) and (i) of regulation 3(2) may, with the approval of the Board, appear in a brochure or leaflet that will accompany the package for the pest control product.
|
| (3) |
Where the information required to be shown on the label is not included in the display panel, the display panel shall contain the words in capital letters "READ ATTACHED BROCHURE BEFORE USE" prominently displayed thereon.
|
|
| 6. |
Display of additional information
| (1) |
Subject to the approval of the Board, additional information relating to the pest control product and any graphic design or symbol maybe shown on the label if it does not unreasonably detract or obscure the information required to be shown on the label under these Regulations.
|
| (2) |
A registrant may include on the label the following liability limitation warranty—
Seller’s guarantee is limited to the terms set out on the label and subject thereto, the buyer assumes the risk to persons or property arising from the use or handling of this product and accepts the product on that condition.
|
|
| 7. |
Information where product is distributed in bulk
Where a pest control product is distributed in a bulk container, the information referred to in subparagraphs (a), (b), (e), (f), (g), (h), (i) and (m) of regulation 3(2) shall be shown—
| (a) |
on the bulk container; and
|
| (b) |
on the documents respecting the pest control product or on a statement accompanying the consignment.
|
|
| 8. |
Information on label
| (1) |
The information on every label shall be printed in both English and Kiswahili languages.
|
| (2) |
All units of measures shown on the label shall be expressed in accordance with the requirements of the Weights and Measures Act (Cap. 513).
|
| (3) |
All information shown on the label shall be printed in a manner that is conspicuous, legible and indelible.
|
|
| 9. |
Denaturation
Where the physical properties of a pest control product are such that the presence of the pest control product may not be recognized when it is used, and is likely to expose a person or domestic animal to a severe health risk, the pest control product shall be denatured by means of colour, odour or such other means as the Board may approve to provide signal or warning of its presence.
|
| 10. |
Storage and display
A pest control product shall be stored and displayed in accordance with the conditions shown on the label.
|
| 11. |
Distribution
Notwithstanding the provisions of the Pharmacy and Poisons Act (Cap. 244), a pest control product shall be distributed in accordance with such condition as the Board shall specify.
|
| 12. |
Prohibition
No person shall use a pest control product in a manner that is inconsistent with the directions or limitations respecting its use as shown on the label.
|
| 13. |
Packaging
| (1) |
Every package for a pest control product shall, unless the Board otherwise directs, be approved by the Board.
|
| (2) |
The package for every pest control product shall be sufficiently durable and be designed and manufactured to contain the pest control product safety under practical conditions of storage, display and distribution.
|
| (3) |
Every package shall be designed and manufactured to permit—
| (a) |
the withdrawal of any or all of the contents in a manner that is safe to the user; and
|
| (b) |
the closing of the package in a manner that will contain the pest control product satisfactorily under practical conditions.
|
|
| (4) |
Every package shall be constructed in such a manner as to minimize the degradation or change of its contents resulting from interaction or from the effects of radiation or other means.
|
|
| 14. |
General prohibitions
| (1) |
A label shall not contain any information respecting any organism or causative agent of a disease that is required to be reported under the Animal Diseases Act (Cap. 364).
|
| (2) |
Information that is required under these Regulations to be shown on a label shall not appear at the bottom of the package.
|
| (3) |
Words stating, implying or inferring that a pest control product is approved, accepted or recommended by the government or by any department or agency thereof shall not appear on a package or label or in any advertisement respecting a pest control product.
|
|
FIRST SCHEDULE [r. 3(2)]
CLASSIFICATION OF PEST CONTROL PRODUCTS
[L.N. 127/2006, rr. 4, 5 and 6.]
| 1. |
Restricted Class
Where the Board has set forth additional essential conditions to be shown on the label respecting the display, distribution, use, limitations or qualifications of persons who may use the pest control product, the pest control product shall have the following properties—
| (a) |
acute oral LD50 is greater than 50 mg/kg body weight;
|
| (b) |
acute dermal LD50 is greater than 100 mg/kg body weight;
|
| (c) |
environmental risks are significant and will be judged accordingly;
|
| (d) |
pest control products used in aquatic and forestry situations are classified restricted.
|
[L.N. 127/2006, rr. 4 and 5.]
|
| 2. |
Commercial and Agricultural Class
Where the pest control product is to be displayed and distributed for general use in commercial activities specified on the label it shall have the following properties—
| (a) |
acute oral LD50 is greater than 50 mg/kg body weight;
|
| (b) |
acute dermal LD50 is greater than 100 mg/kg body weight;
|
| (c) |
environmental effects possible in limited regions.
|
|
| 3. |
Domestic Class
Where the pest control product is to be displayed and distributed for the use in and around a dwelling it shall have the following properties—
| (a) |
acute oral LD50 is over 500 mg/kg body weight;
|
| (b) |
acute dermal LD50 is over 1,000 mg/kg body weight;
|
| (c) |
no special precautions or equipment required for inhalation hazard;
|
| (d) |
no irreversible effects from repeated exposures;
|
| (e) |
disposal of product and containers can safely be done by placing in garbage;
|
| (f) |
package sizes limited to amounts that can be safely used and stored by consumers.
|
|
SECOND SCHEDULE [r. 3(2)]
CAUTIONARY SYMBOLS AND WORDS
[L.N. 127/2006, r. 7.]
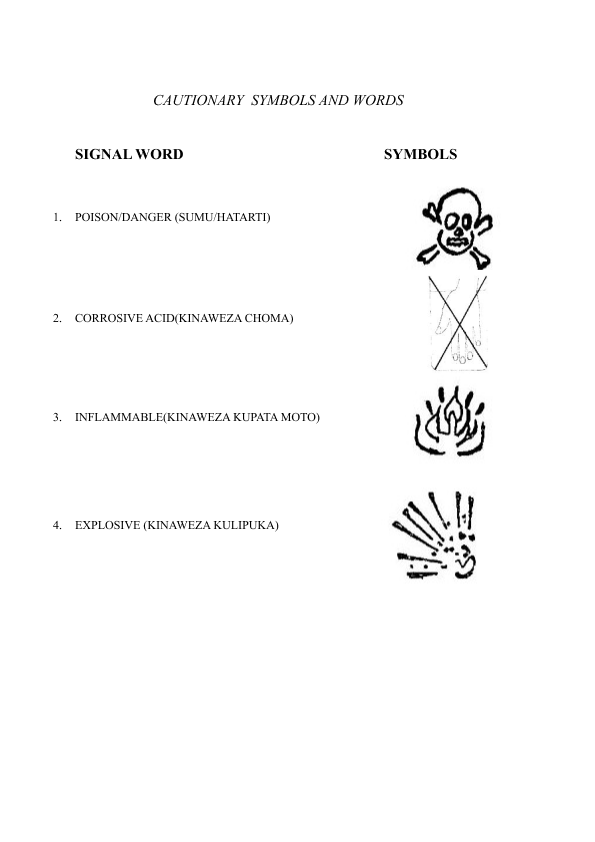
THE PEST CONTROL PRODUCTS (IMPORTATION AND EXPORTATION) REGULATIONS
ARRANGEMENT OF REGULATIONS
| 2. |
Importation and exportation prohibited unless licensed
|
| 4. |
Application for licences
|
| 5. |
Dealing with applications and issue of licences, etc.
|
| 6. |
Duty of licensees, etc.
|
| 7. |
Duty of customs officers
|
| 8. |
Cancellation and suspension of licences
|
| 9. |
Imported pest control products to comply with these Regulations
|
| 10. |
Licence prescribed form
|
SCHEDULES
THE PEST CONTROL PRODUCTS (DISPOSAL) REGULATIONS, 2006
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Disposal) Regulations, 2006.
|
| 2. |
Disposal of pest control products for commercial purpose
| (1) |
No person shall dispose of any pest control product or their containers in a manner that shall be detrimental to man, animal and the environment.
|
| (2) |
No person shall carry out any disposal of pest control product or containers for commercial purposes unless that person or institution is in possession of a licence under these Regulations and all provisions of the Act have been complied with.
|
| (3) |
The use of any pest control product disposal method shall be approved by the Board subject to specific requirements laid down by the Board.
|
| (4) |
The transboundary movements of pesticides for disposal shall be subject to ratified conventions, protocols, treaties or agreements to which Kenya is a party.
|
| (5) |
Without prejudice to the provisions of the Environmental Management and Co-ordination Act, any person or institution desiring a licence for disposal of pest control products for commercial purposes shall apply to the Board for a licence in the prescribed Form A set out in the Schedule and shall be accompanied by the prescribed fees.
|
|
| 3. |
Application for disposal licence
| (1) |
The Board may approve an application made under Regulation 2 and shall issue a licence as set out in Form B in the Schedule if it is satisfied that:
| (a) |
the application contains the information required under these regulations;
|
| (b) |
approved methods of disposal would be used or applied;
|
| (c) |
the applicant has sufficient expertise to dispose of the pest control products in question.
|
|
| (2) |
Disposal for commercial purposes shall be supervised by the Board which shall issue a certificate of disposal in the prescribed Form C in the Schedule after successful disposal.
|
| (3) |
Where the Board does not approve an application it shall not be obliged to give any reason thereof.
|
| (4) |
The Board may attach to a licence such conditions as it deems fit.
|
| (5) |
No person or institution to which a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence to any person or institution without the approval of the Board, which approval shall be endorsed on the licence.
|
|
| 4. |
Cancellation or suspension of licence
| (1) |
The Board may cancel or suspend for a period it deems fit the licence issued under these Regulations if satisfied that-
| (a) |
the licensee, his servant or agent has been convicted of an offence under the Act; or
|
| (b) |
the licensee, his servant or agent has breached any of the terms or conditions of the licence; or
|
| (c) |
for any other reason, that it is in public interest to do so.
|
|
| (2) |
Before cancelling or suspending a licence under this regulation, the Board shall give the licensee twenty-one days' notice to show cause why the licence should not be cancelled or suspended and the Board's decision in the matter shall be final.
|
|
THE PEST CONTROL PRODUCTS (IMPORTATION AND EXPORTATION) REGULATIONS, 1984
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Importation and Exportation) Regulations, 1984.
|
| 2. |
Importation and exportation prohibited unless licensed
| (1) |
No person shall import or export a pest control product for commercial purposes unless that person is in possession of a licence issued under these Regulations.
|
| (2) |
No licence shall be issued for the importation of a pest control product unless the pest control product is registered in accordance with the Pest Control Products (Registration) Regulations, 1984.
|
| (3) |
No licence shall be issued under these Regulations unless the Board is satisfied that the provisions of the Pest Control Products Act, 1982 have been complied with.
|
|
| 3. |
Presumptions
| (1) |
For the purpose of paragraph (1) of regulation 2, a pest control product shall be considered to be for commercial purposes if it is for a use other than scientific research or testing and products imported for resale, manufacture, formulation, owner’s use or commodity aid shall be deemed to be imported for commercial purposes.
|
| (2) |
For the purpose of paragraph (1) of regulation 2, a pest control product shall be deemed to have been exported when it is placed on any ship, aircraft, train or vehicle within Kenya for the purpose of export.
|
|
| 4. |
Application for licences
| (1) |
Any person desiring a licence in respect of importation or exportation of a pest control product for commercial purposes shall apply to the Board for a licence in Form PCPB 1 set out in the Schedule which form shall be signed by the importer or exporter, as the case may be, who shall state the purpose of the importation or exportation of the pest control product in the following manner—
| (i) |
"FOR RESALE", where the product is being imported or exported for the purpose of resale whether in the original pack or after repacking; or |
| (ii) |
"FOR MANUFACTURING PURPOSES", where the pest control product is being imported or exported for use in the manufacture or formulation of a registered pest control product; or |
| (iii) |
"FOR IMPORTER’S OWN USE", where the pest control product is being imported into Kenya or exported to another country for the sole use of the importer, together with information respecting where the pest control product shall be used and the nature of that use. |
|
| (2) |
The Board shall from time to time determine the fee to be paid in respect of an application for a licence.
|
|
| 5. |
Dealing with applications and issue of licences, etc.
| (1) |
The Board may approve an application made under regulation 4 if it is satisfied that—
| (a) |
the application contains the information required under that regulation;
|
| (b) |
the pest control product contains ingredients that have been previously assessed or evaluated for the purposes of the Act and these Regulations and which have been accepted for registration under the Pest Control Products (Registration) Regulations, 1984, for the use stated in the application;
|
| (c) |
premises where the pest control product is to be stored, processed, warehoused, used, repacked or sold shall have been duly licensed by the Board,
|
and issue a licence in Form PCPB 1 set out in the Schedule.
|
| (2) |
Where the Board does not approve an application it shall not be obliged to give any reason therefor.
|
| (3) |
No person to whom a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence to any other person without the approval of the Board, which approval shall be endorsed on the licence.
|
| (4) |
The Board may attach to a licence such conditions as it deems fit to impose and in particular any such condition may restrict the importation and exportation of a pest control product from a specified source or for specified usage and the Board may, from time to time, vary, add to or revoke any of the conditions attached to a licence.
|
| (5) |
The licence issued under regulation 2(1) shall be valid for three (3) months from the date of issue.
[L.N. 125/2006, r. 2.]
|
|
| 6. |
Duty of licensees, etc.
| (1) |
Repealed by L.N. 125/2006, r. 3.
|
| (2) |
It shall be the sole responsibility of the importer or his agent to ensure that Form PCPB 1 is endorsed by the Customs Department at the port of entry and to forward a signed copy to the Board
[L.N. 125/2006, r. 3.]
|
|
| 7. |
Duty of customs officers
Where for any reason a customs officer at a port of entry withholds the release of any pest control product he shall forthwith advise the Board of that action.
|
| 8. |
Cancellation and suspension of licences
| (1) |
The Board may cancel or, for such period as it thinks fit, suspend a licence issued under these Regulations if the Board is satisfied that—
| (a) |
the licensee or his servant or agent has been convicted of an offence under the Act; or
|
| (b) |
the licensee or his servant or agent has committed a breach of any of the terms or conditions of the licence; or
|
| (c) |
for any other reason, it is in the public interest so to do.
|
|
| (2) |
Before cancelling or suspending a licence under this Regulation the Board shall give the licensee twenty-eight days’ notice to show cause why the licence should not be cancelled or suspended and the Board’s decision in the matter shall be final.
|
|
| 9. |
Imported pest control products to comply with these Regulations
No person shall package, store, transport, display, distribute, sell or otherwise deal in a pest control product that has not been imported in accordance with these Regulations.
[L.N. 125/2006, r. 4.]
|
| 10. |
Licence prescribed form
A licence to import or export a pest control product under these Regulations shall be in the prescribed Form PCPB2 set out in the Schedule.
[L.N. 125/2006, r. 5.]
|
THE PEST CONTROL PRODUCTS (IMPORTATION AND EXPORTATION) REGULATIONS
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Importation and Exportation) Regulations.
|
| 2. |
Importation and exportation prohibited unless licensed
| (1) |
No person shall import or export a pest control product for commercial purposes unless that person is in possession of a licence issued under these Regulations.
|
| (2) |
No licence shall be issued for the importation of a pest control product unless the pest control product is registered in accordance with the Pest Control Products (Registration) Regulations.
|
| (3) |
No licence shall be issued under these Regulations unless the Board is satisfied that the provisions of the Pest Control Products Act, 1982 have been complied with.
|
|
| 3. |
Presumptions
| (1) |
For the purpose of paragraph (1) of regulation 2, a pest control product shall be considered to be for commercial purposes if it is for a use other than scientific research or testing and products imported for resale, manufacture, formulation, owner’s use or commodity aid shall be deemed to be imported for commercial purposes.
|
| (2) |
For the purpose of paragraph (1) of regulation 2, a pest control product shall be deemed to have been exported when it is placed on any ship, aircraft, train or vehicle within Kenya for the purpose of export.
|
|
| 4. |
Application for licences
| (1) |
Any person desiring a licence in respect of importation or exportation of a pest control product for commercial purposes shall apply to the Board for a licence in Form PCPB 1 set out in the Schedule which form shall be signed by the importer or exporter, as the case may be, who shall state the purpose of the importation or exportation of the pest control product in the following manner—
| (i) |
"FOR RESALE", where the product is being imported or exported for the purpose of resale whether in the original pack or after repacking; or |
| (ii) |
"FOR MANUFACTURING PURPOSES", where the pest control product is being imported or exported for use in the manufacture or formulation of a registered pest control product; or |
| (iii) |
"FOR IMPORTER’S OWN USE", where the pest control product is being imported into Kenya or exported to another country for the sole use of the importer, together with information respecting where the pest control product shall be used and the nature of that use. |
|
| (2) |
The Board shall from time to time determine the fee to be paid in respect of an application for a licence.
|
|
| 5. |
Dealing with applications and issue of licences, etc.
| (1) |
The Board may approve an application made under regulation 4 if it is satisfied that—
| (a) |
the application contains the information required under that regulation;
|
| (b) |
the pest control product contains ingredients that have been previously assessed or evaluated for the purposes of the Act and these Regulations and which have been accepted for registration under the Pest Control Products (Registration) Regulations, for the use stated in the application;
|
| (c) |
premises where the pest control product is to be stored, processed, warehoused, used, repacked or sold shall have been duly licensed by the Board,
|
and issue a licence in Form PCPB 1 set out in the Schedule.
|
| (2) |
Where the Board does not approve an application it shall not be obliged to give any reason therefor.
|
| (3) |
No person to whom a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence to any other person without the approval of the Board, which approval shall be endorsed on the licence.
|
| (4) |
The Board may attach to a licence such conditions as it deems fit to impose and in particular any such condition may restrict the importation and exportation of a pest control product from a specified source or for specified usage and the Board may, from time to time, vary, add to or revoke any of the conditions attached to a licence.
|
| (5) |
The licence issued under regulation 2(1) shall be valid for three (3) months from the date of issue.
[L.N. 125/2006, r. 2.]
|
|
| 6. |
Duty of licensees, etc.
| (1) |
Repealed by L.N. 125/2006, r. 3.
|
| (2) |
It shall be the sole responsibility of the importer or his agent to ensure that Form PCPB 1 is endorsed by the Customs Department at the port of entry and to forward a signed copy to the Board
[L.N. 125/2006, r. 3.]
|
|
| 7. |
Duty of customs officers
Where for any reason a customs officer at a port of entry withholds the release of any pest control product he shall forthwith advise the Board of that action.
|
| 8. |
Cancellation and suspension of licences
| (1) |
The Board may cancel or, for such period as it thinks fit, suspend a licence issued under these Regulations if the Board is satisfied that—
| (a) |
the licensee or his servant or agent has been convicted of an offence under the Act; or
|
| (b) |
the licensee or his servant or agent has committed a breach of any of the terms or conditions of the licence; or
|
| (c) |
for any other reason, it is in the public interest so to do.
|
|
| (2) |
Before cancelling or suspending a licence under this Regulation the Board shall give the licensee twenty-eight days’ notice to show cause why the licence should not be cancelled or suspended and the Board’s decision in the matter shall be final.
|
|
| 9. |
Imported pest control products to comply with these Regulations
No person shall package, store, transport, display, distribute, sell or otherwise deal in a pest control product that has not been imported in accordance with these Regulations.
[L.N. 125/2006, r. 4.]
|
| 10. |
Licence prescribed form
A licence to import or export a pest control product under these Regulations shall be in the prescribed Form PCPB2 set out in the Schedule.
[L.N. 125/2006, r. 5.]
|
SCHEDULE
|
FORM PCPB 1
|
|
|
THE PEST CONTROL PRODUCTS ACT(Cap. 346)
|
|
PEST CONTROL PRODUCTS (IMPORTANT AND EXPORTATION) REGULATIONS
|
|
APPLICATION NO. .................................
|
|
APPLICATION FOR THE IMPORT/EXPORT OF PEST CONTROL PRODUCTS FOR COMMERCIAL PURPOSE
|
|
(to be typed and submitted in quadruplicate)
|
|
TO:The SecretaryPest Control Products BoardWaiyaki WayP.O Box 13794- 00800NAIROBI.Tel. 254-020-4446115, Fax. 254-020-4449072E-mail: pcpboard@todays.co.keWebsite: www.pcpb.or.ke
|
Applicant's Full Name/Address ..............................................
................................
PIN ...............................................................
Tel.No. ........................................ E-mail .................................
Full name and address of the Exporter (If Import to Kenya) or Importer
(If Export from Kenya) ......................................................
Product Information
| 1. |
Registration number ............................ Date of expiry ...............................
|
| 2. |
Country of origin (if being imported) ...............................
|
| 3. |
Country of destination (if being exported or re-exported) ...........................................
|
| 4. |
SITIC No. ...............................................................
|
| 5. |
Approved common name (a.i.) ...............................................
|
| 6. |
Chemical name ........................................................
|
| 7. |
Trade name ....................................................................
|
| 8. |
Formulation type (EC, EW, Dust, GR, etc.) ..............................................
|
| 9. |
Concentration (% a.i) ...........................................................................
|
| 10. |
State of product (☐ technical or ☐ formulated)
|
| 11. |
| (a) |
Category of product (Tick appropriately)
☐ insecticide ☐ Fungicide ☐ Rodenticide
☐ Acaricide ☐ Herbicide ☐ Adjuvant
Others .......................................
|
| (b) |
Purpose of Import/export ...................
☐ Resale ☐ Manufacturing purposes ☐ Own Use
☐ Trial Samples
Others ....................................................
|
|
| 12. |
Quality applied for ...........................................................
|
| 13. |
Total cost (Freights charges included) ..................... F. O. B. value .......
Currency ................................ Exchange Rate ..........................
Proforma invoice No. .................. Date .........................
|
| 14. |
Estimated annual usage ............................................................
|
| 15. |
Previous imports (gives dates, quality and registration number of products, specify the country of origin) ..........................................................................................
|
PCPB 1/DATE
|
COUNTRY OF ORIGIN
|
QUANTITY
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 16. |
HS Code .............................................................................
|
| 17. |
Custom Entry No. ....................................................................
|
| 18. |
Quantity in stock ...................................................................
I/We certify that the information contained herein is correct to the best of my/our knowledge.
Importer's/Exporter's Signature ........................................................
Designation ............................................................................
Date ....................................................................................
Official stamp ..........................................................................
FOR OFFICIAL USE ONLY
Board's official stamp
|
☐Recommended
|
☐ Not Recommended
|
☐Recommended
|
☐Not Recommended
|
|
Date ............................................
|
Date .............................................
|
|
............................................................Registration Officer
Pest Control Board
|
.................................................Inspector
Pest Control Products Board
|
|
Note; This is not a license to authorize importation or exportation.
|
Fees Paid ...........................................................................
DATE ....................... RECEIPT NO. ............................
|
_________________________
|
FORM PCPB 2
|
|
|
THE PEST CONTROL PRODUCTS (IMPORTATION AND EXPORTATION) REGULATIONS
|
LICENCE NO. .................................................................................
Application No. ................................................................................
This licence is granted to .....................................................................
To import/export a pest control products(s) ........................................................
Trade name .............................................................................
Registration number .......................................................................
Date of expiry of registration ..............................................................
Registred uses ...............................................................................
Country of origin (if being imported) .........................................................
County of destination (if being exported) .......................................................
SITIC No. ..............................................................................
HS Code .................................................................................
Custom Entry No. .......................................................................
Approved Entry No. ......................................................................
Approved common name ......................................................................
Chemical name .............................................................................
Formulation type ..............................................................................
Concentration (a.i %) ..........................................................................
|
State of product (Tick appropriately)
|
☐ Technical or
|
☐ Formulated
|
|
|
Category of product (Tick appropriately)
|
☐ Insecticide
|
☐ Fungicide
|
|
|
|
☐ Herbicide
|
☐ Acaricide
|
☐ Rodenticide
|
|
|
☐ Adjuvant
|
☐ Others ...................................
|
|
|
|
|
Purpose of import/export
☐ Resale ☐Manufacturing Purposes ☐ Own use ☐ Trial Samples
☐ Others...............
Registered use .................................................................
Quantity authorized for importation or exportation ...........................................................
Date .................................................................
|
|
....................................Board's Official Signature and Stamp
|
|
|
.........................................Managing Director, Pest Control Products Board
|
Fee paid .................. Date ................ R. No. ..........................
This license is not transferable to any other person without the approval of the Board.
NB. - This license is valid for one consignment only, for three (3) months from the date of approval.
|
TO BE ENTRED BY THE CUSTOMS OFFICER
|
I certify that the following goods have today been imported or expected.
Trade name ................................................................
Quantity .....................................................................
CIF Value declared ...............................................................
Origin (country and company) ......................................................
Vessel ................................................................................
Clearing agent ....................................................................
Customs entry No. ............................. Date ..................................
Stamp and signature .............................................................
Date ...............................................................................
Notes
| 1. |
Origin to be returned to the Board after customes declaration.
|
| 2. |
Duplicate to be retained by importer/ exporter after customs declaration.
|
| 3. |
Triplicate to be retained by the Board after approval/rejection.
|
THE PEST CONTROL PRODUCTS (DISPOSAL) REGULATIONS
ARRANGEMENT OF REGULATIONS
| 2. |
Disposal of pest control products for commercial purpose
|
| 3. |
Application for disposal licence
|
| 4. |
Cancellation or suspension of licence
|
SCHEDULES
THE PEST CONTROL PRODUCTS (LICENCE FEES AND OTHER CHARGES) REGULATIONS, 2006
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Licence Fees and Other Charges) Regulations, 2006.
|
| 2. |
Fees payable
The fees payable to the Board by any person for services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule and the fees chargeable shall be added in case of combination of businesses in category I in the Schedule.
|
| 3. |
Charges for supervision services
The charges for supervision services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule.
|
| 4. |
Import fee
The Board may charge an import fee at the rate of 0.8% free on board (F.O.B.) value of the product and shall be paid to the Board as agreed with the Agrochemicals Association of Kenya of which 0.4% shall be used by the Board to train end-users and other stake holders on safe use of products and the remaining 0.4% shall be remitted by the Board to the Association for the same purpose of training.
|
SCHEDULE
LICENSING OF PREMISES
THE PEST CONTROL PRODUCTS (DISPOSAL) REGULATIONS, 2006
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Disposal) Regulations, 2006.
|
| 2. |
Disposal of pest control products for commercial purpose
| (1) |
No person shall dispose of any pest control product or their containers in a manner that shall be detrimental to man, animal and the environment.
|
| (2) |
No person shall carry out any disposal of pest control product or containers for commercial purposes unless that person or institution is in possession of a licence under these Regulations and all provisions of the Act have been complied with.
|
| (3) |
The use of any pest control product disposal method shall be approved by the Board subject to specific requirements laid down by the Board.
|
| (4) |
The transboundary movements of pesticides for disposal shall be subject to ratified conventions, protocols, treaties or agreements to which Kenya is a party.
|
| (5) |
Without prejudice to the provisions of the Environmental Management and Co-ordination Act, any person or institution desiring a licence for disposal of pest control products for commercial purposes shall apply to the Board for a licence in the prescribed Form A set out in the Schedule and shall be accompanied by the prescribed fees.
|
|
| 3. |
Application for disposal licence
| (1) |
The Board may approve an application made under Regulation 2 and shall issue a licence as set out in Form B in the Schedule if it is satisfied that:
| (a) |
the application contains the information required under these regulations;
|
| (b) |
approved methods of disposal would be used or applied;
|
| (c) |
the applicant has sufficient expertise to dispose of the pest control products in question.
|
|
| (2) |
Disposal for commercial purposes shall be supervised by the Board which shall issue a certificate of disposal in the prescribed Form C in the Schedule after successful disposal.
|
| (3) |
Where the Board does not approve an application it shall not be obliged to give any reason thereof.
|
| (4) |
The Board may attach to a licence such conditions as it deems fit.
|
| (5) |
No person or institution to which a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence to any person or institution without the approval of the Board, which approval shall be endorsed on the licence.
|
|
| 4. |
Cancellation or suspension of licence
| (1) |
The Board may cancel or suspend for a period it deems fit the licence issued under these Regulations if satisfied that-
| (a) |
the licensee, his servant or agent has been convicted of an offence under the Act; or
|
| (b) |
the licensee, his servant or agent has breached any of the terms or conditions of the licence; or
|
| (c) |
for any other reason, that it is in public interest to do so.
|
|
| (2) |
Before cancelling or suspending a licence under this regulation, the Board shall give the licensee twenty-one days' notice to show cause why the licence should not be cancelled or suspended and the Board's decision in the matter shall be final.
|
|
THE PEST CONTROL PRODUCTS (DISPOSAL) REGULATIONS
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Disposal) Regulations.
|
| 2. |
Disposal of pest control products for commercial purpose
| (1) |
No person shall dispose of any pest control product or their containers in a manner that shall be detrimental to man, animal and the environment.
|
| (2) |
No person shall carry out any disposal of pest control product or containers for commercial purposes unless that person or institution is in possession of a licence under these Regulations and all provisions of the Act have been complied with.
|
| (3) |
The use of any pest control product disposal method shall be approved by the Board subject to specific requirements laid down by the Board.
|
| (4) |
The transboundary movements of pesticides for disposal shall be subject to ratified conventions, protocols, treaties or agreements to which Kenya is a party.
|
| (5) |
Without prejudice to the provisions of the Environmental Management and Co-ordination Act, any person or institution desiring a licence for disposal of pest control products for commercial purposes shall apply to the Board for a licence in the prescribed Form A set out in the Schedule and shall be accompanied by the prescribed fees.
|
|
| 3. |
Application for disposal licence
| (1) |
The Board may approve an application made under Regulation 2 and shall issue a licence as set out in Form B in the Schedule if it is satisfied that:
| (a) |
the application contains the information required under these regulations;
|
| (b) |
approved methods of disposal would be used or applied;
|
| (c) |
the applicant has sufficient expertise to dispose of the pest control products in question.
|
|
| (2) |
Disposal for commercial purposes shall be supervised by the Board which shall issue a certificate of disposal in the prescribed Form C in the Schedule after successful disposal.
|
| (3) |
Where the Board does not approve an application it shall not be obliged to give any reason thereof.
|
| (4) |
The Board may attach to a licence such conditions as it deems fit.
|
| (5) |
No person or institution to which a licence has been issued under these Regulations shall lend, hire, sell, transfer or otherwise dispose of that licence to any person or institution without the approval of the Board, which approval shall be endorsed on the licence.
|
|
| 4. |
Cancellation or suspension of licence
| (1) |
The Board may cancel or suspend for a period it deems fit the licence issued under these Regulations if satisfied that-
| (a) |
the licensee, his servant or agent has been convicted of an offence under the Act; or
|
| (b) |
the licensee, his servant or agent has breached any of the terms or conditions of the licence; or
|
| (c) |
for any other reason, that it is in public interest to do so.
|
|
| (2) |
Before cancelling or suspending a licence under this regulation, the Board shall give the licensee twenty-one days' notice to show cause why the licence should not be cancelled or suspended and the Board's decision in the matter shall be final.
|
|
SCHEDULE
FORMS
|
FORM A
|
|
THE PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
APPLICATION FOR THE DISPOSAL OF PEST CONTROL PRODUCTS FOR COMMERCIAL PURPOSE
|
|
(to be typed or printed)
|
To: The Managing Director,
Pest Control Product Board,
P.O.BOX 13794-00800,
Westlands-Nairobi.
1. Applicant's full Name/ Address ................................................................
................................................................................................................
2. The company directors ................................................................
....................................................................................................
3. Types of waste/(products, formulations, state etc.)..............................................
4. Quality in details ....................................................................................
5. Source(s) of wastes ...................................................................................
6. Reasons for disposal ................................................................................
7. Methods of disposal to be used .................................................................
(attach the details or the methods)
8. Qualifications and experience of personnel ..........................................................
9. Disposal site ..................................... (attach details) .......................................
10. Transportation to the disposal site ................................................................
...................................................................................................................
11. Environmental impact Assessment Report .........................................................
|
Dated ...........................Sign/Stamp.................
|
Date .............................
|
|
|
.......................................Signature of applicant
|
___________________________________
|
FORM B
|
|
|
THE PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
THE PEST CONTROL PRODUCT DISPOSAL REGULATIONS
|
LICENCE NO. .........................................................
This licence is granted to ................................................
to dispose the following pest control products wastes ................................. using the following methods ................................ at ..................................
|
Dated ......................................Signature ..............................
|
Managing Director pest control products Board
|
|
|
....................................Signature of applicant
|
This licence is not transferable to any of the person or institution.
___________________________________
|
FORM C
|
|
|
THE PEST CONTROL PRODUCTS ACT[Cap. 346]
|
|
THE PEST CONTROL PRODUCT DISPOSAL REGULATIONS
|
CERTIFICATE OF DISPOSAL NO. ............................................
This is to certify that on. ........................................................
satisfactorily disposed of the following pest control products:
1. ........................................................................................................
2. ......................................................................................................
3. .......................................................................................................
4. .......................................................................................................
5. .......................................................................................................
Methods of disposal:
1. .........................................................................................................
2. .........................................................................................................
3. ........................................................................................................
4. ........................................................................................................
Comments from the Board ...............................................................
............................................................................................................
|
.......................................Managing Director
|
.......................................Date
|
Stamp ..................................................
The disposal certificate is not transferable.
THE PEST CONTROL PRODUCTS (LICENCE FEES AND OTHER CHARGES) REGULATIONS
ARRANGEMENT OF REGULATIONS
| 3. |
Charges for supervision services
|
SCHEDULES
| SCHEDULE — |
LICENSING OF PREMISES
|
THE PEST CONTROL PRODUCTS (LICENCE FEES AND OTHER CHARGES) REGULATIONS, 2006
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Licence Fees and Other Charges) Regulations, 2006.
|
| 2. |
Fees payable
The fees payable to the Board by any person for services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule and the fees chargeable shall be added in case of combination of businesses in category I in the Schedule.
|
| 3. |
Charges for supervision services
The charges for supervision services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule.
|
| 4. |
Import fee
The Board may charge an import fee at the rate of 0.8% free on board (F.O.B.) value of the product and shall be paid to the Board as agreed with the Agrochemicals Association of Kenya of which 0.4% shall be used by the Board to train end-users and other stake holders on safe use of products and the remaining 0.4% shall be remitted by the Board to the Association for the same purpose of training.
|
SCHEDULE
LICENSING OF PREMISES
THE PEST CONTROL PRODUCTS (LICENCE FEES AND OTHER CHARGES) REGULATIONS
| 1. |
Citation
These Regulations may be cited as the Pest Control Products (Licence Fees and Other Charges) Regulations.
|
| 2. |
Fees payable
The fees payable to the Board by any person for services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule and the fees chargeable shall be added in case of combination of businesses in category I in the Schedule.
|
| 3. |
Charges for supervision services
The charges for supervision services rendered by the Board shall be as prescribed by the Board from time to time as set out in the Schedule.
|
| 4. |
Import fee
The Board may charge an import fee at the rate of 0.8% free on board (F.O.B.) value of the product and shall be paid to the Board as agreed with the Agrochemicals Association of Kenya of which 0.4% shall be used by the Board to train end-users and other stake holders on safe use of products and the remaining 0.4% shall be remitted by the Board to the Association for the same purpose of training.
|
SCHEDULE
LICENSING OF PREMISES
|
Category
|
Annual Licence Fees (Ksh.)
|
|
(a) Retail
|
1,000
|
|
(b) Supermarkets
|
2,000
|
|
(c) Distribution/Wholesale/Agents/Importers and other business dealing with pest control products not covered under (a)-(g)
|
4,000
|
|
(d) (Re-packing)
|
5,000
|
|
(e) Formulation/manufacture
|
7,500
|
|
(f) Commercial Pest Control Operators
|
2,000
|
|
(g) Storage/ Warehousing and Use
|
5,000
|
|
(h) Change of Agent (per product)
|
20,000
|
|
DISPOSAL OF PEST CONTROL PRODUCTS
|
|
(a) Licence for disposal of waste
|
5,000
|
|
(b) Charges for supervision
|
5% of the cost of disposal.
|
|
REGISTRATION OF PEST CONTROL PRODUCTS(Per product)
|
|
(a) Certificate of Registration (3 years)
|
30,000
|
|
(b) Renewal of Certificate of Registration not exceeding 2 years
|
20,000
|
|
(c) Temporary Registration for a period not exceeding 1 year
|
10,000
|
|
(d) Introduction of new products (experimental permit)
|
10,000
|
|
IMPORT AND EXPORT OF PEST CONTROL PRODUCTS(Per product)
|
|
(a) Imports of less than Kshs. 500,000 in value
|
2,000
|
|
(b) Imports of Ksh. 500,000 and over in value
|
0.8% of F.O.B. value
|
|
(c) Exports of any amount
|
1,000
|
|